Case Study
Vibroacoustic Therapy for Ehlers-Danlos Syndrome: A Case Study
Picard LM1,2*, Bartel LR1, Gordon AS1,2, Denise Paneduro2, Sally Chung2 and Pink LR2
1Music and Health Research Collaboratory, University of Toronto, USA
2Wasser Pain Management Centre, Mount Sinai Hospital, Toronto Ontario, USA
*Corresponding author: Larry M Picard, Music and Health Research Collaboratory, Wasser Pain Management Centre, Mount Sinai Hospital, University of Toronto, USA
Published: 01 Mar, 2018
Cite this article as: Picard LM, Bartel LR, Gordon AS,
Paneduro D, Chung S, Pink LR.
Vibroacoustic Therapy for Ehlers-
Danlos Syndrome: A Case Study. Ann
Clin Case Rep. 2018; 3: 1504.
Abstract
Background: Widespread pain and tenderness are prominent features of Ehlers-Danlos Syndrome
Hypermobility Type (EDS-HT). Chronic pain in this condition may be perpetuated by central
nervous system sensitization and is refractory to treatment. Vibroacoustic therapy has the potential
to modulate pain responses via somatosensory input.
Objective: To examine the effects of a short course of self-administered, home-based vibroacoustic
chair therapy as add-on treatment in EDS.
Methods: A patient with painful EDS was supplied with a vibroacoustic chair. The patient used the
chair up to seven days per week twice per day for 53 out of a possible 58 sessions over 29 consecutive
treatment days (approximately 4 weeks), followed by a two week “washout” period. Responses were
assessed using the Brief Pain Inventory, Short Form (BPI-SF) and the Sleep Quality Numerical
Rating Scale (SQNRS) administered at study initiation, and at the end of weeks 2, 4 and 6. A patient
global impression of change (GIC-P) was obtained at the conclusion of active treatment (week 4).
Thirty minute semi-structured interviews were carried out at the end of week 4 and week 6. The
change from baseline in the BPI subscales, the Pain Severity Score (PSS) and the Pain Interference
Score (PIS), from baseline to end of the active treatment period and patient global impression of
change were the primary end points. Changes in the SQNRS over the course of the study and the
changes in BPI subscales and from time of treatment termination until the final assessment were the
secondary endpoints.
Results: The PSS was 7.75 at baseline and unchanged at termination of treatment. The PIS
improved by 2.43 points from baseline (8.86) to termination of active treatment (6.43). Patient
Global Impression of Change was “slightly better” following treatment. The patient experienced a
mostly positive stimulation of bowel function, relaxation and/or lessening of muscle tension, and an
improvement in energy levels. Bothersome tingling sensations were experienced especially close to
the time of initiation for a brief period but there were no serious adverse effects. During the washout
period, the PSS increased slightly by 0.25 points between the termination of therapy (7.75) and final
follow-up (8.0). The PIS changed 1.14 points, from 6.43 at termination of therapy to 7.57 at final
assessment. Sleep quality also worsened during the washout period with an increase in the SQNRS
scale from 8 to 9 although it did not change from baseline during the active treatment period. There
was some overall deterioration in symptom control in the two weeks after treatment termination.
Conclusions: This N of 1 study suggests modest symptom improvement from vibroacoustic therapy
in EDS-HT. There was an unexpected effect of bowel stimulation. Larger controlled studies are
needed to confirm and extend these findings.
Introduction
The Ehlers-Danlos Syndromes (EDS) are a heterogeneous group of heritable connective
tissue disorders characterized by articular hypermobility, skin extensibility, and tissue fragility.
Widespread pain and tenderness are prominent features of the autosomal dominant Ehlers–Danlos
syndrome hypermobility type (EDS-HT). Major diagnostic criteria include skin involvement with
hyperextensibility and/or smooth, velvety skin as well as generalized joint hypermobility.Joint
hypermobility is the dominant clinical manifestation with certain joints, having a propensity to
dislocate[1].
It has been postulated that early in the course of the condition mechanical factors account for the pain but later central nervous system sensitization produces
generalized hyperalgesia[2,3]. While pain predominates, depression,
fatigue, impaired sleep, and diminished quality of life occur [4].
The pain in this condition is refractory to treatment [5].
Vibroacoustic therapy (VAT) uses sound and mechanical
vibrations in the audible range applied directly to the body with
therapeutic intent [6]. The mechanisms of action responsible for
improvement in these areas may involve stimulation of Pacinian
corpuscles, direct mechanical tissue stimulation, induction of the
relaxation response and cellular/subcellular effects. Mechanical
stimulation in the range of 60-600 Hz activates Pacinian corpuscles,
which are involved in somatosensation and may influence pain
perception [7-10]. At least some of the benefits may derive from
reduced muscle tension [11].
VAT has produced significant gains in the relief of pain of
musculoskeletal origin [12,13], including fibromyalgia and back pain
[14,15].
Dr. Patrick of the NIH Clinical Center has treated more than
15,000 patients having a wide variety of medical conditions [16].
Skille and other therapists have data from more than 40,000 hours of
treatment. No serious adverse reactions have occurred [17].
Given the potential for benefit, the relative safety of VAT and the
lack of alternative therapies, we considered that a trial of VAT in a
patient diagnosed with EDS-HT was justified.
Materials and Methods
Participant
Diagnosis: The patient was a 19-year-old male student of
Ashkenazi Jewish and Polish/Russian background. He carried a
diagnosis of Ehlers-Danlos syndrome (EDS) spectrum disorder of the
hypermobile type with features of classical type.
Pain onset and symptoms: The patient had the onset of
widespread pain prior to his 14th birthday. At that time, he developed
flu-like symptoms consisting of a sore throat, abdominal pain,
vomiting, fever, cough, night sweats and generalized aching. He was
initially treated with a three day course of clarithromycin for suspected
pneumonia but a chest x-ray proved normal. The most remarkable
feature of a detailed workup was an antistreptolysin O titer of 402.
Principal ongoing pain complaints were focused on the lower limbs
and only occasionally in the hands. There were intermittent sharp
pains superimposed on long-duration pain. The patient rated his
baseline pain as eight out of 10. He was unable to walk without a cane
and had given up sporting activities.
Medical history: Going back to childhood there were recurrent
shoulder and carpal-metacarpal joint dislocations as well as knee
buckling. Additionally, he experienced urinary frequency and
urgency, depressive symptoms, anxiety, panic attacks and sleep
disturbances. Additional medical history included bilateral inguinal
hernias at approximately age 10 and a small periumbilical hernia. The
inguinal hernias were treated surgically. He was noted to have soft
stretchable skin without any skin fragility or easy bruising. There had
been no spontaneous skin tearing.
Family history: A brother, seven years older, maternal first cousin
and mother’s paternal uncle had suffered from similar skin disorders
and pain. Another brother, three years older and a sister, three years
younger had similar skin but no pain complaints.
Clinical examination: The patient’s Beighton score was five out
of nine confirming hypermobility. There wereno pseudotumors or
piezogenic papules. Brisk deep tendon reflexes at the knees and some
brief clonus at the ankles of uncertain significance were noted but no
other neurological abnormalities were documented.
Investigations: A skin punch biopsy contained numerous
scattered dysmorphic fibrils including many flower-like collagen
fibrils with moderate diameter variability. There were no other
significant findings by light and electron microscopy and elastin
fibers were normal. Genetic testing was extensive but he was known
to have a COL5A1 variant, 190 3C >T translocation.
Sleep studies demonstrated arousals at four minute intervals
and decreased REM sleep. He had extensive imaging with MRI,
including the brain, the knees and proximal muscles. All the studies
were negative or normal. MRI studies of the length of the spine
showed only a small (incidental) Tarlov cyst at T10-T11 which was
radiographically stable for 4 years. An echocardiogram was negative
for mitral regurgitation and mitral valve prolapse. The aorta and
aortic root appeared normal. A bladder ultrasound, done while he
was experiencing a sense of fullness, was associated with only 4 mL
of fluid.
Attempted therapies: Sleep hygiene and massage provided some
benefit. Physiotherapy improved his shoulder strength. Interventions
without substantial benefit included relaxation techniques and
medications of varying types. These included tramadol containing
medications and opioids, cannabinoids, mirtazapine, dopamine
receptor blockers, "Z drugs", baclofen, gabapentinoids and SNRI's.
Inclusion and Exclusion Criteria
Contraindications to the use of VAT were reviewed (Table 1) and determined to be absent before the trial of therapy was initiated [18]. Discussion with our patient confirmed that he had adequate English proficiency for completion of questionnaires, the ability to tolerate the treatment and a willingness to complete the study assessments.
Intervention(VAT)
The study was of six weeks’ duration; with four weeks of active
treatment followed by two weeks’ “washout.” It was intended that the
patient receive 56 treatment sessions given as two sessions per day
over 28 consecutive days. However, the actual usage was 53 sessions
over 29 consecutive days. Treatment was self-administered with
a vibroacoustic chair (Sound Oasis Model VTS-1000, Sound Oasis
Company, Marblehead MA 01945-0526, USA).
The first session of the day included 30 minutes of the Sound
Oasis “Energize” program consisting of three music tracks with a
component of low frequency gamma entrainment (41 Hz to 73 Hz
with 41 Hz dominant; 36 Hz to 61 Hz with 41 Hz dominant; and
36 Hz to 65 Hz with 41 Hz dominant) and with mono and binaural
high alpha (10Hz) and beta (15Hz) entrainment. The patient was
instructed to play the “Energize” program at an intensity level of 15
and a volume level of 1 or 2. The second session of the day included
30 minutes of the Sound Oasis “Sleep” program consisting of three
music tracks with a component of low frequency gamma entrainment
(32 Hz; range between 27.5 Hz to 44 Hz; and range between 34 Hz to
52 Hz) and with mono and binaural delta (2Hz) entrainment. The
patient was instructed to play the “Sleep” program at an intensity
level of 5 and a volume level of 1 or 2.
The patient was given specific instructions (both in person and
via a supplied manual) regarding usage of the VAT system. The
research coordinator was available to answer any questions. In the
event of adverse reactions the patient was advised to discontinue
the intervention and report the reaction to one of the study team
members.
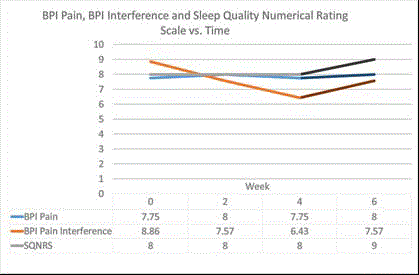
Figure 1
Figure 1
The three measures compatible with a less favorable clinical state
two weeks following termination of active treatment.
Measures
The Brief Pain Inventory (Short Form; BPI-SF,Copyright 1991
Charles S. Cleeland, PhD, used with permission) [19] was employed
to measure the severity of patient’s pain and pain-related functional
impairment.The pain severity score (PSS) is calculated by obtaining a
mean score from 4 questions that patients report, rating the severity
of their pain at its “worst”, “least”, “average”, and “now”. These
responses are measured on an 11-point scale from 0 (no pain) to 10
(pain as bad as you can imagine). Pain interference is also measured
on an 11-point scale and assesses the degree to which pain interferes
in 7 categories including: daily activities, general activity, walking,
work, mood, enjoyment of life, relations with others and sleep. A
mean score is obtained for these 7 items (PIS). Reliability of the PSS
and PIS is good as indicated by alpha coefficients of 0.85 and 0.88,
respectively [20].
Sleep Quality Numerical Rating Scale (SQNRS). Sleep quality was
measured on an 11 point Likert scale with 0 corresponding to best
possible sleep and 10 to worst possible sleep was administered [21].
Patient Global Impression of Change (PGIC): A patient global
impression of change was collected on a 7 point Likert scale (range 1
=Very much worse, 2= Much worse, 3= slightly worse, 4= No change,
5= slightly better, 6= Much better, 7= Very much better).
Assessments
At the first study visit, written informed consent and consent
to the transmission of personal health information via email was
obtained. Since the study involved only one participant, Research
Ethics Board approval was not required. The trial was conducted in
accordance with the principals set out in the Declaration of Helsinki
[22]. The BPI-SF and SQNRS were administered at study initiation
and at the end of weeks 2, 4 and 6.The PGIC was administered at
the end of active treatment (week 4). The patient was given the
vibroacoustic therapy system to take home for the duration of active
therapy. The patient was contacted by the research coordinator two
days following the first visit to ensure he was comfortable using the
VAT system.
Forms for data collection were provided to the patient at study
initiation.Dates and duration of usage of the study intervention, and
the presence or absence of side-effects were recorded by the patient
on a calendar. The patient was provided with specific instructions
(in person and via instruction manual) regarding how to complete
the calendar and was asked to submit the completed calendar via
email weekly. The research coordinator contacted the patient weekly
to ensure completion and submission of the calendar. The patient
was instructed to present himself in person for the completion of
assessments, or if this was not possible, to communicate the content
of the forms electronically via email. For the first three visits (baseline,
weeks 2 and 4), the patient completed the forms in person. The final
assessment forms (week 6) were completed at home and sent to the
research coordinator via email. There were three telephone treatment
check-ins, the first occurred 2 days following the baseline visit, the
second occurred one week following the baseline visit and the last
occurred at week 6.
Thirty minute semi-structured interviews took place in person
at week 4, following active treatment, and over the phone at week
6, following the 2 week washout period. The goal was to explore the
patient’s experience and perceived effectiveness of the VAT system
for managing pain and sleep. The participant was asked about any
new treatments started and any changes in medication.
Statistical Analysis
The change from baseline in the PSS and PIS to end of the active treatment period and the PGIC were the primary end points. Secondary endpoints were changes in the SQNRS over the course of the study and changes in the PSS, PIS and from time of treatment termination until the final assessment. Data were managed and results plotted in Microsoft Excel 2010 (Microsoft, Redmond, Washington USA).
Results
Primary findings
The BPI pain severity score measured 7.75 at baseline and was
unchanged at termination of treatment. The BPI pain interference
score improved by 2.43 points from baseline (8.86) to termination
of active treatment (6.43). Following active treatment, the patient
assigned a value of 5 on the PGIC scale, corresponding to “slightly
better” indicating the patient perceived an improvement in his
condition.
Secondary findings
The PSS increased slightly by 0.25 points between the termination
of therapy (7.75) and the final follow-up two weeks later (8.0). The
PIS increased 1.14 points, from 6.43 at termination of therapy to 7.57
at final assessment, indicating a worsening in pain disability. Sleep
quality also worsened during this timeframe reflected in an increase
on the SQNRS from 8 to 9. However, the SQNRS (8) remained
unchanged from baseline over the course of active treatment.
Therefore, all three measures were compatible with a less favorable
clinical state two weeks following termination of active treatment.
Figure 1 illustrates these findings.
Freeform Observations
The patient made freeform observations on 9 out of 29 (31%)
treatment days and on 11 out of 15 (73%) post-treatment days. These subjective responses will be reported together with responses from
the semi-structured interviews at week four and week six.
The patient likened the experience of using the chair to having a
massage and felt it would substitute for one.
The patient’s use of the chair was extensive.He used it seven
days per week and for 53 out of a possible 58 sessions. During the
treatment phases there were consistent and/or repeated reports that
VAT had significant activating effects on bowel function.Relaxation
and/or lessening of muscle tension was reported on four days. The
patient remarked that the treatment was relaxing but also helped to
mobilize him in the morning.
He described an improvement in energy levels. There appeared to
be effects on productivity which in turn elevated his mood. However,
he could not be sure that the chair, rather than physiotherapy and
additional self-help measures brought about these improvements.
The patient only made one change to his medication regime
during the treatment period one day prior to termination. At that
time he began taking a medication to counter an overactive bladder.
He also began more challenging physiotherapy exercises during the
treatment period,focusing on strengthening the shoulder and bladder.
In the post-treatment phase the patient reported increased muscle
tightness or pain, headaches, sleep problems and he also reported
increasing his bladder mediation dose from 4mg to 8mg/night. The
medication dose increase was probably not linked to the termination
of active treatment.
With respect to reported side effects, the patient experienced
tingling sensations especially close to the time of initiating therapy.
The tingling was described as “bothersome.”In addition, effects
of chills and nausea were reported once each on treatment days.
While the patient felt that the use of the chair had mostly positive
effects on his bowels, sometimes the stimulating effects led to a short
interruption of treatment, which he found mildly bothersome.
Commenting on the shortcomings of the system, the patient
noted that the chair did not touch his legs and therefore did not benefit
that part of the body. Furthermore, the discrepancies in contours of
the chair and of the patient’s back diminished the comfort of the
treatment. He did find the time commitment to be an additional
drawback.
At the termination of active treatment (week 4) the patient was
asked to respond to four specific questions and to rate his response
from 0, representing not helpful at all to 10, representing extremely
helpful.The questions and responses are summarized in table 2.
Following the 2-week washout period, in retrospect he found
the VAT to be helpful for increasing his energy levels and providing
a restful sleep, which in turn, impacted his mood positively. He
reported that the increase in energy and improved mood encouraged
him to take an important step and seek out a volunteer opportunity,
something he felt unable to do prior to treatment due to his level of
pain severity and interference.The patient remained unsure whether
the chair or other factors fully accounted for the changes which
occurred during the study timeframe. However, post-washout that
he now believed the VAT to be mostly responsible for these changes.
Given these positive experience, he indicated that he would
choose to use the system in the future and in fact approached us for
information as to how he could acquire a chair for personal use. It was
his preference to use the chair prior to sleep and on an “as needed”
basis rather than twice per day.
Table 1
Table 2
Discussion
Use of the vibroacoustic chair was associated with some decline
in the pain interference score over the four week treatment interval.
However, there was no detectable change in the pain severity score
or sleep quality from initiation to termination of active treatment.
The patient’s global impression of change was positive but only
moderately so.
The pain interference score showed some worsening of pain in
the washout period. There was also a decline in sleep quality at the
final assessment. Although minimal, the BPI pain severity score also
showed worsening at the same time point.
Taken together with the patient’s narrative reports, these finding
are suggestive of very modest treatment effects accruing during
the active phase of therapy and declining thereafter. There were
no significant adverse effects but bothersome tingling especially in
the early treatment sessions which could conceivably limit patient
acceptance of the treatment in the event that it proves to be a common
rather than isolated experience.
Given that this is an N of 1 unblinded study the significance of
the reported findings is difficult to interpret. As this was an add-on
therapy, it is not possible to attribute all of the changes seen during
the study period to the effects of the chair. In addition to purely
placebo effects, the other concurrently applied interventions and
random variation may have contributed to the outcomes.
The noticeable lack of effect on the lower limbs, which would not
have received direct stimulation in this treatment paradigm, is also
of interest. Our expectation was that stimulation of the torso would
produce a generalized benefit extending beyond the site of application.
The study patient’s ongoing pain complaints were concentrated in
the lower limbs. It is possible that patients’ whose pain lies is largely
centered in the torso might show more robust treatment effects.
Regardless of these limitations, the patient’s positive responses
have to be gauged against the refractory nature of his condition.
VAT requires further study for the treatment of painful Ehlers-
Danlos and other chronic pain disorders such as fibromyalgia. The
effect of bowel activation was unexpected and suggests that VAT
of the torso might be beneficial for conditions are associated with
chronic constipation.
Equipment manufactures should consider designing consumer
VAT units that provides wide area stimulation including the lower
limbs as well as the torso.
Conclusion
VAT offers an innovative, treatment approach that can be selfadministered with few side effects in patients with EDS. Further research is necessary to define its role in therapy.
References
- Beighton P, De Paepe A, Steinmann B, Tsipouras P, Wenstrup RJ. Ehlers-Danlos syndromes: revised nosology, Villefranche, 1997. Ehlers-Danlos National Foundation (USA) and Ehlers-Danlos Support Group (UK). Am J Med Genet. 1998;77(1):31-7.
- Rombaut L, Scheper M, De Wandele I, De Vries J, Meeus M, Malfait F, et al. Chronic pain in patients with the hypermobility type of Ehlers-Danlos syndrome: evidence for generalized hyperalgesia. Clin Rheumatol. 2015;34(6):1121-9.
- Castori M, Morlino S, Celletti C, Ghibellini G, Bruschini M, Grammatico P, et al. Re-writing the natural history of pain and related symptoms in the joint hypermobility syndrome/Ehlers-Danlos syndrome, hypermobility type. Am J Med Genet A. 2013;161A(12):2989-3004.
- Albayrak I, Yilmaz H, Akkurt HE, Salli A, Karaca G. Is pain the only symptom in patients with benign joint hypermobility syndrome?. Clin Rheumatol. 2015;34(9):1613-9.
- Rombaut L, Malfait F, De Wandele I, Cools A, Thijs Y, De Paepe A, et al.. Medication, surgery, and physiotherapy among patients with the hypermobility type of Ehlers-Danlos syndrome. Arch Phys Med Rehabil. 2011;92(7):1106-12.
- Hooper J. An introduction to vibroacoustic therapy and an examination of its place in music therapy practice. Br J Music Ther. 2001;15(2):69-77.
- Chesky KS, Michel DE, Kondraske G. Developing methods and techniques for scientific and medical application of music vibration. In: Spintge R, Droh R, editors. Music Medicine. Vol. II. St. Louis: MMB Music; 1996.
- Quillian TA, Sato M. The distribution of myelin and nerve fibres from Pacinian Corpuscles. J Physiol. 1955;129(1):167-76.
- Hubbard SJ. A study of rapid mechanical events in a mechanoreceptor. J Physiol 1958;141(2):198-218.1.
- Lundenberg T, Nordemarr R, Ottoson D. Pain alleviation by vibratory stimulation. Pain. 1984;20(1):25-44.
- Skille 0. Manual of vibroacoustic therapy. Levanger, Norway: ISVA.
- Patrick G. The effects of vibroacoustic music on symptom reduction: Inducing the relaxation response through good vibrations. lEE Eng Med Biol Mag. 1999;18(2):97-100.
- Michel DE, Chesky K. Music and music vibration for pain relief: Standards in research. In: Spintge R, Dron R, editors. Music Medicine. Vol. 2. St. Louis: MMB Music; 1996. p. 218-226.
- Wigram T. The effects of vibroacoustic therapy on clinical and non-clinical populations [doctoral dissertation, St. George's Medical School, London University 1996]. Online document at: quadrillo.tripod.com/ -quadrillo/index-4.html.
- Naghdi L, Ahonen H, Macario P, Bartel L. The effect of low-frequency sound stimulation on patients with fibromyalgia: a clinical study. Pain Res Manag. 2015;20(1):21-7.
- Patrick G. The effects of vibroacoustic music on symptom reduction. IEE Eng Med Biol Mag. 1999;18:97-100.
- Boyd-Brewer C, McCaffrey R. Vibroacoustic sound therapy improves pain management and more. Holist Nurs Pract. 2004;18(1):111-8.
- Grocke DE, Wigram T. Receptive methods in music therapy: Techniques and clinical applications for music therapy clinicians, educators and students. London & Philadelphia: Jessica Kingsley; 2007. Chapter 9, Vibroacoustic Therapy in Receptive Music Therapy. 214-235.
- Brief Pain Inventory (BPI). The University of Texas MD Anderson Cancer Center.
- Tan G, Jensen MP, Thornby JI, Shanti BF. Validation of the Brief Pain Inventory for chronic nonmalignant pain. J Pain. 2004;5(2):133-7.
- Martin S, Chandran A, Zografos L, Zlateva G. Evaluation of the impact of fibromyalgia on patients' sleep and the content validity of two sleep scales. Health Qual Life Outcomes. 2009;7:64.
- World Medical Association. Ethics Unit. Declaration of Helsinki. 2013.