Case Report
Liver Manifestation of Atypical Hemolytic Uremic Syndrome Associated with Heterozygous Polymorphism C-332T>C in the Promoter of the Complement Factor H Gene - Case Report
Dorota Brodowska-Kania1, Aleksandra Paturej1, Ewa Kotwica-Strzałek1*, Marek Saracyn1, Michał Nowicki2 and Stanisław Niemczyk1
1Department of Internal Medicine, Nephrology and Dialysis, Military Institute of Medicine, Poland
2Department of Nephrology, Hypertension and Kidney Transplantation, Medical University of Lodz, Poland
*Corresponding author: Ewa Kotwica-Strzałek, Department of Internal Medicine, Nephrology and Dialysis, Military Institute of Medicine, Warsaw Szaserów 128, 01-141 Warsaw, Poland
Published: 30 Jun, 2018
Cite this article as: Brodowska-Kania D, Paturej A,
Kotwica-Strzałek E, Saracyn M, Nowicki
M, Niemczyk S. Liver Manifestation of
Atypical Hemolytic Uremic Syndrome
Associated with Heterozygous
Polymorphism C-332T>C in the
Promoter of the Complement Factor
H Gene - Case Report. Ann Clin Case
Rep. 2018; 3: 1529.
Abstract
Background: Primary atypical hemolytic uremic syndrome (aHUS) is caused by complement
dysregulation. Diagnosis is based on determining the presence of hemolytic anemia,
thrombocytopenia, and the presence of schistocytes in the peripheral blood smear and damage
to internal organs - most commonly the kidneys. Liver involvement as the main symptom of the
disease is rare, making it difficult to recognize.
Case Presentation: We present a case of a 65 year old woman whose clinical picture was dominated
by weakness, vomiting, jaundice, disturbances of consciousness, as well as characteristics of liver
and kidney damage. Laboratory tests showed typical features of hemolytic anemia Hgb 9.3g%,
high LDH, low haptoglobin, thrombocytopenia 116x109/l, signs of renal damage: creatinine
3.6mg/dl, and elevated bilirubin level 5.5mg/dl. Manual differential blood smear stated 6% to
8% schistocytes. Establishing a diagnosis was possible after genetic testing, which showed only a
heterozygous polymorphism in the promoter of the factor H gene. Eculizumab was not an option
(lack of registration in Poland), thus other methods of treatment were sought (infusions of plasma,
plasmapheresis and corticosteroids).
Conclusion: Atypical hemolytic uremic syndrome is not always acute. For carriers of the mutated
gene, the image of the disease may be incomplete and precede in a chronic way that delays diagnosis.
Hepatic manifestation of aHUS is casuistic in medicine and probably results from damage to the
vascular endothelial cells in small hepatic vessels.
Keywords: Thrombotic microangiopathy; Atypical hemolytic-uremic syndrome; Complement
factor H mutation; Case report
Abbreviations
aHUS: Atypical Hemolitic-Uremic Syndrome; STEC-HUS: Diarrhea-Associated Hemolytic Uremic Syndrome; TTP: Thrombotic Thrombocytopenic Purpura; HGB: Hemoglobin; HCT: Hematocrit; RBC: Red Blood Cell Count; ALT: Alanine Transaminase; AST: Aspartate Transaminase; PLT: Platelets; INR: International Normalized Ratio; DD: Dimer D; ATIII: Antithrombin III; anti-LKM1: Anti-Liver-Kidney Microsome 1; ANCA: Anti-Neutrophil Cytoplasmic Antibody; ANA: Antinuclear Antibody; SMA: Smooth Muscle Antibody; antidsDNA- anti-double stranded DNA; MRI: Magnetic Resonance Imaging; PET: Positron Emission Tomography; LDH: Lactate Dehydrogenase; CC3: Complement Component 3; FFP: Fresh Frozen Plasma; anti-MASP2; CFH: Complement Factor H; MCP: Membrane Cofactor Protein; CFI: Complement Factor I; CFB: Complement Factor B; THBD: Thrombomodulin; ADAMTS 13: Disintegrin and Metalloproteinase with a thrombospondin type 1 motif; member 13; MLDPA: Multiplex Ligation-Dependent Probe Amplification; DGKE: Diacylglycerol Kinase ε; SLE: Systemic Lupus Erythromatosus; HELLP: Syndrome Hemolytic Anemia; Elevated Liver Enzymes; Low Platelet Count; DIC: Disseminated Intravascular Coagulation; TMA: Thrombotic Microangiopathies; anti-CFH: Autoantibodies against Complement Factor H; EMA: European Medicine Agency
Background
Atypical hemolytic uremic syndrome (aHUS) is a rare disease associated with excessive complement activation [1,2,3]. Subsequent endothelial damage and disseminate coagulation lead to hemolytic anemia and thrombocytopenia. These symptoms lie at the center of thrombotic microangiopathies: aHUS, diarrhea-associated hemolytic uremic syndrome (STEC-HUS) and thrombotic thrombocytopenic purpura (TTP). Eculizumab, a monoclonal antibody against compliment component 5, is the new standard of care for aHUS. As eculizumab was not registered in Poland during patient’s hospitalization, other methods of treatment were sought (plasma infusion, plasma exchange and glicocortycosteroids). Following case of aHUS is unique for the predominance of liver symptoms and the chronic clinical course.
Presentation Case
We present a case of a 65-year old woman, Caucasian race, body
weight of 56 kg, who was admitted to Tertiary Nephrologic Center
with postprandial vomiting, recurrent vertigo and general asthenia.
Clinical examination revealed xanthochromia and liver enlargement
(5 cm beneath the ribcage). Three years prior to first hospitalization
a routine examination during a surgical provision of left arm fracture
revealed a bilirubin level of 2 mg/dl. No chronic diseases in patient
medical history. At the age of 31 patients gave birth to a healthy
daughter, with no complications during pregnancy and childbirth.
During present hospitalization laboratory tests showed: anemia
(hemoglobin HGB 9.3g/l, hematocrit HCT 27%, red blood cell
count RBC 2.84x1012/l), kidney and liver dysfunction (creatinine
3.6 mg/dl, urea 72 mg/dl, bilirubin 5.5 mg/dl, alanine transaminase
ALT 20 U/l, aspartate transaminase AST 46 U/l), moderate
thrombocytopenia (platelets PLT 116x109/l) and the following
coagulation disorders: prolonged international normalized ratio
(INR), an increased D-Dimer and a decreased antithrombin 3 value
(ATIII). Abdominal ultrasonography revealed an oval focal lesion in
the left liver lobe (decreased echogenicity, 14 mm in diameter) and
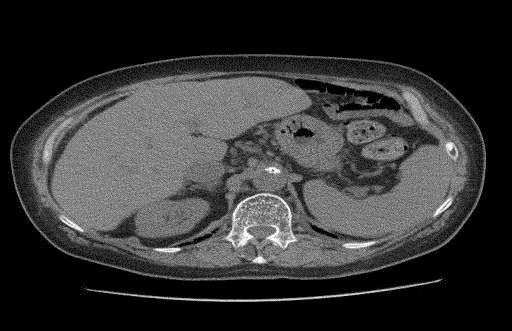
numerous cysts in the right liver lobe. In CT scan, in both lobes of
the liver visible several outbreaks of ~8mm in diameter. Liver was
slightly enlarged, with a reduced coefficient of extinction (approx. 45
J.H.), Figure 1. During the diagnostic process no biopsy or magnetic
resonance imaging (MRI) was conducted due to thrombocytopenia
and the presence of a metal plate in the left arm, Figure 2. A whole
body Positron Emission Tomography (PET) showed no intensified
glucose uptake. Tests for viral and autoimmune hepatitis and
other autoimmune disorders were carried out (for systemic lupus
erythematosus, dermatomyositis, antiphospholipid syndrome,
systemic vasculitis): anti-double stranded DNA (anti-dsDNA)
antibodies, lupus anticoagulant, Antinuclear Antibody (ANA),
anti-liver-kidney microsome 1 (anti-LKM1), Anti-Neutrophil
Cytoplasmic Antibody (ANCA), Smooth Muscle Antibody (SMA).
No antibodies were found. Moreover, the patient suffered from
recurrent episodes of impaired consciousness during hospitalization.
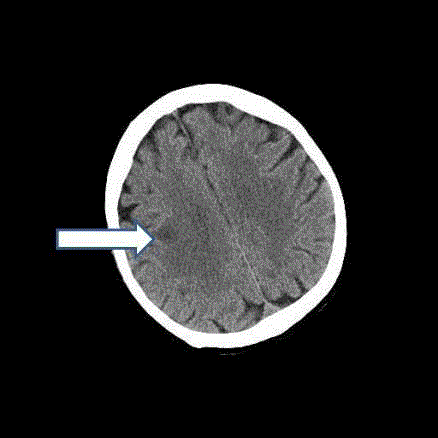
A computer tomography of the head visualized a fresh cerebral
ischemia in the right cerebral hemisphere, Figure 3. Furthermore,
despite blood transfusions anemia and thrombocytopenia intensified.
The presence of schistocytes in the blood smear (6% to 8%), an
increased level of Lactate Dehydrogenase (LDH) and reticulocyte
count and an almost undetectable level of heptoglobin suggested
hemolytic microangiopathic anaemia. Considering the entire clinical
picture, a diagnosis of atypical hemolytic uremic syndrome (aHUS)
or thrombotic thrombocytopenic purpura TTP of unknown origin
was considered. Blood serum was collected and showed a decreased
complement component 3 (CC3) activity 80 mg/dl (90 to 180); and
normal ADAMTS13 activity – 93% (N 40% to 130%). Shiga toxin
was negative. Because of the deteriorating clinical condition, therapy
was started. The infusions of 500 mg methylprednisolone for 3
consecutive days were followed by 40 mg of oral prednisone bringing
a temporary improvement. Additionally, a total of 24 units of Fresh
Frozen Plasma (FFP) were infused. The clinical state improved,
without renal replacement therapy a diuresis of over 2 liters per day
was achieved with a slight deterioration of liver function.
The patient was repeatedly hospitalized for impaired
consciousness accompanied by anemia and a decline of kidney
and liver function manifested by hepatic encephalopathy and
dehydration. The patient received repeated therapeutic plasma
exchanges. Three plasmaphereses were performed, each exchanging
4 liters of plasma with FFP bringing no improvement to the clinical
state. Due to the ineffectiveness of treatment (plasmapheresis,
steroids, FFP transfusion), recurrent relapses of aHUS and lack of
targeted therapy with eculizumab, (not available in Poland at that
time), the patient was offered a participation in a phase 2 clinical
trial examining the safety of the monoclonal antibody anti-MASP 2
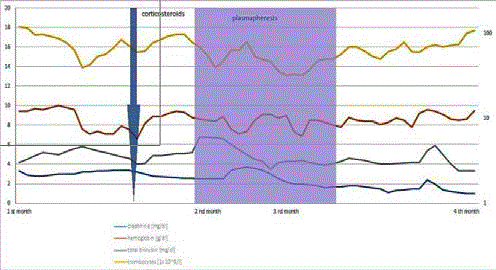
(OMS721-TMA-001, NCT02222545). Change in levels of creatinine,
haemoglobin, bilirubin
and platelets over time are shown on Figure 4. Genetic testing
was performed to assess the most common mutations associated
with HUS/TTP development (Table 1). Testing was performed in
The Clinical Research Center for Rare Diseases “Aldo e CeleDacco”,
Mario Negri Institute for Pharmacological Research, in Italy. No mutations were found by Next Generation Sequencing (NGS)
of Complement Factor H (CFH), Membrane Cofactor Protein
(MCP), Complement Component 3 (CC3), Complement Factor I
(CFI), Complement Factor B (CFB), Thrombomodulin (THBD),
a disintegrin and metalloproteinase with a thrombospondin type 1
motif, member 13 (ADAMTS13), diacylglycerol kinase ε (DGKE)
genes. A heterozygous polymorphism c-332T>C in the promoter
of complement factor H gene was found by Multiplex Ligation-
Dependent Probe Amplification (MLPA). The mutation is associated
with aHUS development.
Figure 1
Figure 1
CT scan. In both lobes of the liver visible several outbreaks
diameter of about 8 mm - cystis?other? In addition, in the segment IVB
vaguely demarcated, low-hyper density change focal - 9 mm. Liver slightly
enlarged, with a reduced coefficient of extinction (approx. 45 J.H.).
Figure 2
Figure 3
Figure 3
CT scan. A computer tomography of the head visualized a fresh
cerebral ischemia in the right cerebral hemisphere.
Figure 4
Discussion
Atypical hemolytic uremic syndrome is a rare disease with
a prevalence of 2.1 cases per million [4-6]. Various complement
abnormalities and excessive coagulation lead to consumptive
thrombocytopenia. Hemolytic anemia is caused by damage to
erythrocytes caused on their way through the narrowed vessels. The
disease is associated with renal function impairment: 60% of children
and 80% of adults eventually require renal replacement therapy
[7]. Neurological (impaired consciousness, stroke, focal deficits
and epileptic seizures) and cardiac involvement is present in 10%
to 30% of cases [5,8]. The main symptom in our patient was liver
malfunction, renal involvement. Hepatic manifestation of aHUS may
have been a result of hemolysis in the endothelium of hepatic vessels
and subsequent organ ischaemia. Only about half of the patients with
typical presentation of aHUS have identifiable complement factor
mutations. Our patient had no mutation of complement factor H but
was heterozygous for the polymorphism c.-332C>T in the promoter
of CFH gene. Such abnormality has already been identified in other
patients with aHUS [9]. Patient was a carrier of the mutated gene, and
this could be a reason for a milder and more chronic course of disease
and subsequent difficulty in establishing diagnosis of aHUS. In order
for the syndrome to develop a combination of factors must coincide:
a mutation, predisposing haplotype and a triggering factor [10].
Around 50% of aHUS cases are preceded with a gastrointestinal or
upper respiratory tract infection [5,11,12,13]. Other triggering events
include pregnancy, cancer and use of medications [14]. No associated
trigger was found in the presented case.
Diagnosis of aHUS must be differentiated with other thrombotic
microangiopathies. Hemolytic anemia, thrombocytopenia and acute
renal insufficiency, fever and a possible central nervous system
involvement are associated with HUS in the course of E. coli O157
infection. No gastroenteritis was present in the described case
and Shiga toxin in stool was negative. Neurological involvement
accompanied by fever, thrombocytopenia, hemolytic anemia and
renal insufficiency is characteristic for TTP caused by low activity of ADAMTS13. In this case ADAMTS13 activity was normal.
Furthermore, thrombotic microangiopathies must be differentiated
with Systemic Lupus Erythromatosus (SLE), syndrome of Hemolytic
Anemia, Elevated Liver Enzymes, Low Platelet Count (HELLP),
Disseminated Intravascular Coagulation (DIC) and Thrombotic
Microangiopathies (TMA) secondary to drugs like quinine,
simvastatin, interferon or calcineurin inhibitors.
If plasmapheresis, is unavailable then FFP transfusion remains
the first line of aHUS treatment. The treatment provides the lacking
complement factors [15]. Exchange of 1-2 plasma volumes in each
plasmapheresis is recommended as was the case in our patient [16].
Immunosuppressive therapy has a limited utility in the treatment of
aHUS showing some effect only in cases related to the development
of autoantibodies against complement factor H anti-CFH [17].
However, FFP infusions, 3 therapeutic plasma exchanges and steroids
provided only a temporary improvement.
In the 2009 European Medicine Agency (EMA) approved
eculizumab for aHUS therapy. It is a monoclonal antibody against C5
complement factor. Its use is highly effective with 80% of the patients
achieving long term remission, as opposed to 5% of patients treated
with plasmaphereses [18]. Due to lack of approval eculizumab was
unavailable in Poland at that time. That was the reason the patient
started experimental therapy as a part of phase 2 clinical trial.
Table 1
Conclusion
Liver manifestation of aHUS is rare. Final diagnosis was established after thorough, extensive diagnostics including genetic testing. Treatment was ineffective, thus other methods of treatment were sought. The main limitation of our manuscript was the inability to treat with eculizumab at that time.
Summary
The above described case presents the difficulty of diagnosing an atypical clinical course of aHUS. Treatment of aHUS presents an even greater challenge, especially if plasma exchanges are ineffective. It is advisable to search for new genes associated with aHUS/TTP. In justified cases, specific diagnosis is possible only after genetic testing, especially for a long period of illness when the symptoms are not spectacular. aHUS is rare syndrome with poor prognosis. Approximately 70% of patients die; it requires dialysis or developing chronic kidney disease within a year after diagnosis [19-20]. It is necessary to search for new therapeutic possibilities.
References
- Buddles MR, Donne RL, Richards A, Goodship J, Goodship Th. Complement factor H gene mutation associated with autosomal recessive atypical hemolytic uremic syndrome. Am J Hum Genet. 2000;66(5):1721-2.
- Wong EKS, Goodship THJ, Kavanagh D. Complement therapy in atypical haemolyticuraemic syndrome (aHUS). MolImmunol. 2013;56(3):199-212.
- Desch K, Motto DJ. Is there a shared pathophysiology for thrombotic thrombocytopenicpurpura and hemolytic-uremic syndrome?. Am Soc Nephrol. 2007;18(9):2457-60.
- Miller D, Kaye J, Shea K, Ziyadeh N, Cali C, Black C, et al. Incidence of Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome. Epidemiology. 2004;15(2):208-15.
- Noris M, Caprioli J Bresin E, Mossali C, Pianetti G, Gamba S, Daina E, et al. Relative role of genetic complement abnormalities in sporadic and familial aHUS and their impact on clinical phenotype. Clin J Am SocNephrol. 2010;5(10):1844-59.
- Maga TK, Nishimura CJ, Weaver AE, Frees KL, Smith RJ. Mutations in alternative pathway complement proteins in American patients with atypical hemolytic uremic syndrome. Hum Mutat. 2010;31(6):E1445-60.
- Fremeaux-Bacchi V, Fakhouri F, Garnier A, Bienaimé F, Dragon-Durey MA, Ngo S, et al. Genetics and outcome of atypical hemolytic uremic syndrome: a nationwide French series comparing children and adults. Clin J Am SocNephrol. 2013;8(4):554-62.
- Ažukaitis K, Loirat C, Malina M, Adomaitienė I, Jankauskienė A. Macrovascular involvement in a child with atypical hemolytic uremic syndrome. PediatrNephrol. 2014;29(7):1273-7.
- Caprioli J, Castelletti F, Bucchioni S, Bettinaglio P, Bresin E, Pianetti G, et al. Complement factor H mutations and gene polymorphisms in haemolyticuraemic syndrome: the C-257T, the A2089G and the G2881T polymorphisms are strongly associated with the disease. Hum Mol Genet. 200;12(24):3385-95.
- Scully M, Hunt BJ, Benjamin S, Liesner R, Rose P, Peyvandi F, etal. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. B J Haemotol. 2012;158(3):323-35.
- Sellier-Leclerc AL, Fremeaux-Bacchi V, Dragon-Durey MA, Macher MA, Niaudet P, Guest G, et al. Differential Impact of Complement Mutations on Clinical Characteristics in Atypical Hemolytic Uremic Syndrome. JAmSocNephrol. 2007;18:2392-400.
- Geerdink LM, Westra D, van Wijk JA, Dorresteijn EM, Lilien MR, Davin JC, et al. Atypical hemolytic uremic syndrome in children: complement mutations and clinical characteristics. PediatrNephrol. 2012;27(8):1283-91.
- Fakhouri F, Roumenina L, Provot F, Sallée M, Caillard S, Couzi L, et al. Pregnancy-Associated Hemolytic Uremic Syndrome Revisited in the Era of Complement Gene Mutations. J Am SocNephrol. 2010;21(5):859-67.
- Kavanagh D, Goodship T, Richards A. Atypical Hemolytic Uremic Syndrome Semin Nephrol. 2013;33(6):508-30.
- Ariceta G, Besbas N, Johnson S, Karpman D, Landau D, Licht C, et al. Guideline for the investigation and initial therapy of diarrhea-negative hemolytic uremic syndrome. Pediatr Nephrol. 2009;24(4):687-96.
- Kavanagh D, Raman S, Sheerin NS. Management of hemolytic uremic syndrome. F1000Prime Rep. 2014;6:119.
- Sinha A, Gulati A, Saini S, Blanc C, Gupta A, Gurjar BS, et al. Prompt plasma exchanges and immunosuppressive treatment improves the outcomes of anti-factor H autoantibody-associated hemolytic uremic syndrome in children. Kidney Int. 2014;85(5):1151-60.
- Legendre CM, Licht C, Muus P, Greenbaum LA, Babu S, Bedrosian C, et al. Terminal complement inhibitor eculizumab in atypical hemolytic–uremic syndrome. N Engl J Med. 2013;368(23):2169-81.
- Fakhouri F, Hourmant M, Campistol JM, Cataland SR, Espinosa M, Gaber AO, et al. Terminal complement inhibitor eculizumab in adult patients with atypical hemolytic uremic syndrome: a single-arm, open-label trial. Am J Kidney Dis. 2016;68(1):84-93.
- Noris M, Remuzzi G. Atypical hemolytic-uremic syndrome. N Engl J Med. 2009;361(17):1676-87.
- Caprioli J, Noris M, Brioschi S, Pianetti G, Castelletti F, Bettinaglio P, et al. Genetics of HUS: the impact of MCP, CFH, and IF mutations on clinical presentation, response to treatment, and outcome. Blood. 2006;108(4):1267-79.