Case Report
Therapeutic Iron Infusion Causing Delayed Venous Contamination in Magnetic Resonance Angiography
Wei Li*, Ioannis Koktzoglou, Eugene Dunkle and Robert R Edelman
Department of Radiology, North Shore University Health System, USA
*Corresponding author: Wei Li, Department of Radiology/ CAI, Walgreen Building, Suite G507, Evanston Hospital, 2650 Ridge Ave, Evanston, IL 60201, USA
Published: 27 Jan, 2018
Cite this article as: Li W, Koktzoglou I, Dunkle E, Edelman
RR. Therapeutic Iron Infusion Causing
Delayed Venous Contamination in
Magnetic Resonance Angiography. Ann
Clin Case Rep. 2018; 3: 1497.
Abstract
The diagnostic quality of magnetic resonance angiography (MRA) depends on the reliable suppression of venous signal, which otherwise overlaps arterial signal. In the case presented here, the quality of non-enhanced MRA was severely contaminated by venous signal due to a therapeutic iron infusion (ferumoxytol, intravascular half-life ~15 hours) two days prior to the MRA exam. The cause of the venous contamination was that intravenously injected ferumoxytol substantially shortened the longitudinal relaxation time (T1) of the blood, so that venous presaturation no longer was effective. This case highlights the importance of considering prior iron infusion as a cause of MRA artifacts and including it in the pre-exam questionnaire.
Keywords: Enhanced MRA; Venous contamination; Feraheme; Ferumoxytol; Iron
Introduction
Diagnostic quality of peripheral magnetic resonance angiography (MRA) depends on the reliable suppression of venous signal, which otherwise overlaps arterial signal. The efficacy of venous suppression relies on the presumption of a long T1 relaxation time for blood, typically on the order of 1.2 – 1.4 sec at 1.5 T [1]. With contrast-enhanced MRA, venous suppression is obtained through the rapid repetition of moderate flip angle radiofrequency pulses. With unenhanced MRA using quiescent-interval single-shot (QISS) or two-dimensional (2D) time-of-flight approaches, venous suppression is obtained through the application of tracking presaturation radio frequency (RF) pulses [2-4]. However, we recently encountered a case in which extensive venous enhancement was encountered due to an iron infusion well prior to the unenhanced MRA exam. The institutional review board of our institution has waived written informed consent from the patient for publication of the case report.
Case Presentation
A 73-year-old female patient with claudication was scheduled for nonenhanced MRA of her
lower extremities because of impaired renal function (estimated glomerular filtration rate (eGFR)
< 30 mL/min/1.73 m2). QISS MRA was performed on a 1.5-T MR scanner (MAGNETOM Avanto;
Siemens Healthcare, Erlangen, Germany) using multiple phased array coils to cover the area from
the level of renal arteries to the feet. Nine-station QISS imaging (48 axial slices per station; 432 total
slices) was prescribed. The major parameters of the QISS sequence included electrocardiographic
(ECG) gating, repetition time (TR) = 3.0 ms echo time (TE) = 1.4 ms, flip angle = 90°, quiescent
interval = 350 ms, trigger delay relative to the R-wave of 100 ms, slice thickness = 3.0 mm, field of
view = 400 ms. Details regarding the imaging protocol can be found in reference [2].
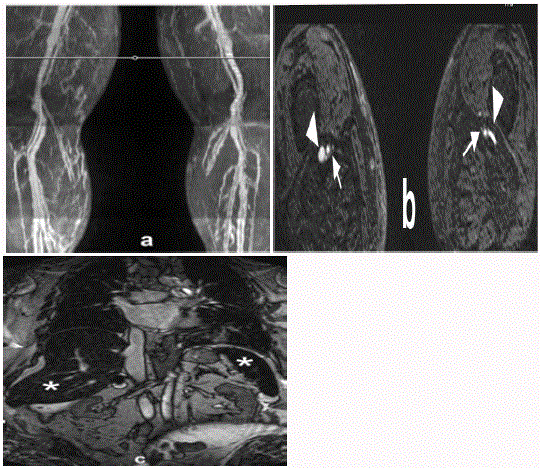
While performing the exam, we observed severe venous contamination (Figure 1A and B). By
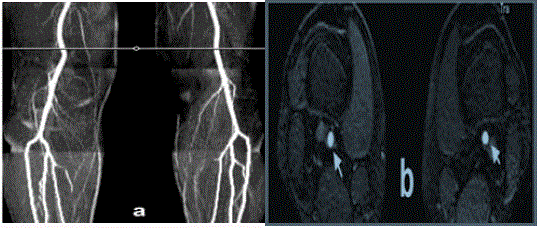
comparison, a QISS MRA performed one day earlier on a healthy volunteer showed clear depiction
of the arteries and excellent suppression of veins and background tissues (Figure 2A and B). To rule out the possibility of scanner dysfunction, we interrupted the study, restarted the MR scanner,
and then repeated the exam which showed no change. Subsequently, on a coronal abdominal scout
image, we observed anomalously low signal from the liver and spleen (Figure 1C). Upon further questioning of the patient, it was discovered that the patient had received an intravenous injection
of 510 mg (30 mg/mL) of Feraheme™ (ferumoxytol, Advanced Magnetics, Cambridge, MA) two days
prior to the MR exam as treatment for iron deficiency anemia.
Figure 1
Figure 1
A 73-year-old female with claudication, poor renal function,
and history of iron infusion two days earlier for treatment of iron deficiency
anemia: a) Coronal maximum intensity projection image from QISS MRA
shows both arteries and veins. b) Transverse source image at the level of the
white horizontal line in (a); both arteries (arrows) and veins (arrowheads) can
be seen. c) Coronal scout image of the abdomen (obtained with a balanced
steady-state free precession sequence) shows low signal from the liver and
spleen (*), indicating iron deposition.
Figure 2
Figure 2
QISS MRA of a 48-year-old male healthy volunteer acquired
using identical imaging parameters to the patient study shown in Figure
1: a) Coronal QISS maximum intensity projection image at the level of
the popliteal trifurcation. Note the excellent depiction of arteries with near
complete suppression of venous signal. b) Transverse QISS source image
at the level of the white horizontal line in (a); the arteries (arrow) are shown
without substantial venous signal.
Discussion
Both QISS and 2D time-of-flight MRA use RF presaturation to suppress the appearance of venous signal. Ferumoxytol infusion is a recently approved therapy
for iron deficiency anemia. It is an iron-based therapy that consists of
ultra-small superparamagnetic particles. Given its safety in patients
with severe renal functional impairment, long intravascular half-life, and ability to shorten the T1 relaxation time of blood, it has also been
tested as an intravascular MRA contrast agent [5-6]. Unlike most
gadolinium-based extracellular chelates, which have an intravascular
half-life on the order of minutes, ferumoxytol has a much longer
intravascular half-life (~15 hours) and shortens the T1 relaxation
time for many hours following administration. Alteration of liver
signal with conventional MR imaging may persist for several months.
Given the frequency of renal functional impairment in patients
with peripheral vascular disease [7] and the associated common
complication of iron deficiency anemia with chronic kidney disease,
information about iron supplements should be included in the
MRI pre-exam questionnaire. If possible, the MRA study should be
conducted prior to the iron agent administration or at least several
days afterwards to avoid the confounding effects of intravascular T1
shortening.
Conclusion
Therapeutic iron infusion should be considered as a potential cause of venous contamination when performing MRA.
References
- Stanisz GJ, Odrobina EE, Pun J, Escaravage M, Graham SJ, Bronskill MJ, et al. T1, T2 relaxation and magnetization transfer in tissue at 3T. Magn Reson Med. 2005; 54: 507-512.
- Edelman RR, Sheehan JJ, Dunkle E, Schindler N, Carr J, Koktzoglou I. Quiescent-interval single-shot unenhanced magnetic resonance angiography of peripheral vascular disease: Technical considerations and clinical feasibility. Magn Reson Med. 2010; 63:951-958.
- Timothy G Scanlon , Jeremy D Collins, John J Sheehan, Eugene E Dunkle, Navyash Gupta, James C Carr, et al. Evaluation of peripheral arterial disease with nonenhanced quiescent-interval single-shot MR angiography. Radiology. 2011; 260: 282-293.
- Leiner T. Magnetic resonance angiography of abdominal and lower extremity vasculature. Top Magn Reson Imaging. 2005; 16: 21-66.
- Li W, Tutton S, Vu AT, Pierchala L, Li BS, Lewis JM, et al. First-pass contrast-enhanced magnetic resonance angiography in humans using ferumoxytol, a novel USPIO-based blood pool agent. J Magn Reson Imaging. 2005; 21: 46-52.
- Li W, Salanitri J, Tutton S, Dunkle EE, Schneider JR, Caprini JA, et al. Lower extremity deep venous thrombosis: evaluation with ferumoxytol-enhanced MR imaging and dual-contrast mechanism—preliminary experience. Radiology. 2007; 242: 873-881.
- Guerrero A, Montes R, Muñoz-Terol J, Gil-Peralta A, Toro J, Naranjo M, et al. Peripheral arterial disease in patients with stages IV and V chronic renal failure. Nephrol Dial Transplant. 2006; 21: 3525-3531.