Clinical Image
Synchronous Primary Tumors of the Liver and Gallbladder: Case Report and Review of the Literature
Kurt J Knowles1*, Lubnia Alattia1, Minyu Cheng1, Quyen Chu2, Elba A Turbat-Herrera1 and Guillermo A Herrera1
1Department of Pathology and Translational Pathobiology, LSU Health Science Center, USA
2Department of Surgery, LSU Health Science Center, USA
*Corresponding author: Kurt J Knowles, Department of Pathology and Translational Pathobiology, LSU Health Science Center, Shreveport Louisiana, 71130, USA
Published: 31 Jul, 2017
Cite this article as: Knowles KJ, Alattia L, Cheng M, Chu
Q, Turbat-Herrera EA, Herrera GA.
Synchronous Primary Tumors of the
Liver and Gallbladder: Case Report and
Review of the Literature. Ann Clin Case
Rep. 2017; 2: 1407.
Abstract
As the longevity of human’s increase and with scientific medical advances the occurrence of multiple primary malignant tumors (MPMT’s) will rise. Synchronous multiple primary malignant tumors are defined as the occurrence of two primary neoplasms within six months of each other. Overall, the occurrence of MPMT’s varies from 1%-16%, with HCC and multiple other tumors very rare and the synchronous occurrence of HCC and gallbladder carcinoma has only been reported 6 times. It is crucial to patient care that each tumor is identified correctly as they are treated entirely different and the prognosis for the patient is dependent on the stage for each.
Introduction
The occurrence of multiple synchronous primary tumors in patients with HCC is extremely rare
and the association HCC and GBC has only been previously reported 6 times.
Criteria for the classification of multiple primary tumors: 1) each tumor is unequivocally
malignant 2) each tumor is a separate and distinct entity 3) must exclude that one is a metastasis
from the other [1]. It is critical that simultaneous tumors be identified as therapy must be guided taking into consideration both tumors and the treatment for each independently, as well as the
treatment for both combined. A thorough review of the literature in PubMed was performed and
articles relevant to this case report cited.
Case Presentation
An elderly male was referred to our institution after computerized tomography (CT) scan
revealed a suspicious mass in the dome of the gallbladder and he was scheduled for a laparoscopic
versus open cholecystectomy with possible radical cholecystectomy. No hepatic lesions were noted
on CT. At the time of the laparoscopic surgery the surgeons were unable to completely visualize
the gallbladder secondary to dense adhesions therefore an open cholecystectomy was performed.
After careful dissection, the gallbladder was isolated and a firm mass was palpated in the fundus.
The node of Calot was dissected and sent to pathology for an intraoperative frozen section which
was positive for carcinoma. A radical cholecystectomy was performed which included additional
lymph node dissection and resection of segment 4 of the liver. The procedure and postoperative care
of the patient was excellent and there were no complications. The patients care was transferred to
a different facility but he was stable one year later with no definitive recurrence of the two tumors.
The gross specimens consisted of a partial hepatectomy 5 cm x 4.5 cm x 4 cm with a poorly
circumscribed 2.5 cm tan yellow mass in the background of a cirrhotic liver. The gallbladder
consisted of a 10 cm x 6 cm x 4 cm gallbladder with a mass consisting of multiple fragments of
friable red papillary tissue in aggregate 4 cm x 3 cm x 2.5 cm. Microscopically the HCC consisted
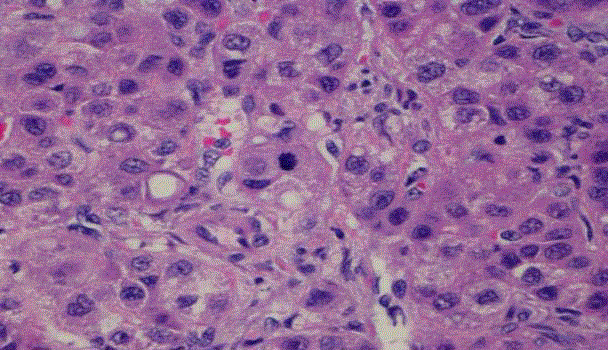
of thickened trabeculae (some 8-10 cells thick) comprised of moderately atypical hepatocytes with
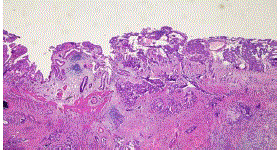
prominent nucleoli, nuclear pseudoinclusions and mitoses (Figure 1). The gallbladder consisted of a moderately differentiated adenocarcinoma that predominantly formed glands although there are
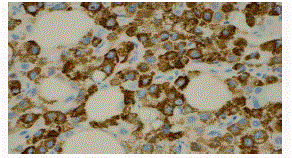
nests of cells infiltrating the gallbladder wall (Figure 2). The GBC was positive for CEA (Figure 3),
CK7, CK20 while the HCC was negative for these markers. The HCC was intensly positive for AFP.
Figure 1
Figure 1
Hepatocellular carcinoma with thickened trabecula, pleomorphic
hepatocytes, mitosis in the center.
Figure 2
Figure 3
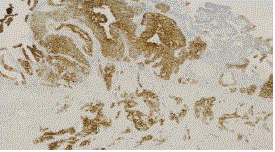
Figure 4
Discussion
As the longevity of human’s increases and medical science capabilities improve the occurrence of multiple primary malignant tumors (MPMT’s) will also increase. There are three established criteria for the classification of multiple primary tumors: (1) each tumor is
unequivocally malignant (2) each tumor is a separate and distinct
entity (3) must exclude that one is a metastasis from the other. These
criteria were first put forth by Warren and Gates in 1932 and for
the most part still remain valid [1]. With today’s modern immunohistochemistry
and genetic analysis the latter two criteria are easily
met and mutually exclusive. Synchronous tumors are defined as
tumors that occur within 6 months of each other and metachronous
tumors occur greater than 6 months of each other. In the present case
the hepatocellular carcinoma (HCC) and the gallbladder carcinoma
(GBC) occurred simultaneously, there was no clinical or histological
connection between the two and IHC demonstrated that the two
tumors are phenotypically distinct and thus one is not a metastasis
from the other (Table 1).
The occurrence of MPMT’s varies considerably, between 0.734%
and 11.7% as documented by Demandante et al. [2] in a literature
review of 1,104,269 patients with MPMT’s and the incidence of
synchronous MPMT’s is rare. Bagri reported that out of 23,260
patients, 41 had MPMT’s and only 9 were synchronous (< 0.04%) [3].
Papaconstantinou et al. [4] reported on a twelve year period and the
incidence was 0.5%, 39 out of 7516. An in-depth and thorough SEER report stated the incidence varied depending on the site of the initial primary and was 1% for liver to 16% for bladder [5].
The occurrence of additional multiple primary tumors in patients
with HCC are extremely rare and the association HCC and GBC has
only been previously reported 6 times. De Pangher Manzini et al. [6]
reported that over a fifteen year period from 1980-1995 there were 29
cases of HCC and MPMT’s, and in two of these cases the additional
tumor was GBC. Wei et al. [7] reported on a series of patients who had
HCC and the associated MPMT’s. Over a 26 year period 448 HCC’s
were identified, 40 had MPMT’s, only 11 had synchronous tumors
(about 2.5%) and there was a single case of HCC and GBC (0.22%).
Imada et al. [8] reported on a case of triple carcinomas; HCC, GBC
and cholangiocarcinoma. Kim et al. [9] reported on a case in which a
patient underwent a partial hepatectomy and at the time of surgery an
intraoperative frozen section discovered an adenocarcinoma of the
gallbladder and La Greca et al. [10] discovered a GBC after resection
for HCC.
Although simultaneous HCC and GBC is extremely rare it
is imperative that they both are identified as the treatment and
prognosisis radically different for each one. The basic concepts
involved in treating a patient with both HCC/GBC can be applied
to any patient with MPMT’s, ie whether surgery, chemotherapy,
immunotherapy or radiation is the desired treatment. For example,
transplant or partial hepatectomy are the primary treatment options
for HCC with the greatest chance of success. However, less than 30%
of GBC is amenable to surgical resection [11] which greatly limits
surgery as a treatment option in HCC. Cytotoxic chemotherapy such
as gemcitabine is used in GBC but chemotherapy is generally not
beneficial in HCC [12,13]. Sorafenib, an inhibitor of the mitogenactivated
pathway, is the only approved chemotherapy agent in HCC
however it is potent bone marrow suppressor and even if it was not
harmful in GBC, the addition of cytotoxic chemotherapy with the
immunologic would be especially harmful to the bone marrow.
Although extremely rare concurrent HCC and GBC are treated
entirely different and their occurrence’s should be recognized
pathologically and clinically. This case superbly demonstrates the
multifaceted aspects of treating patients with multiple primary tumors
and as the longevity of human’s increase, and with the capabilities
of the medical profession rapidly advancing, multiple primary malignant tumors will probably become more commonplace.
References
- Warren S, Gates O. Multiple primary malignant tumors: a survey of the literature and statistical study. Am J Cancer. 1932; 16: 1358-1414.
- Demandante CG, Troyer DA, Miles TP. Multiple primary malignant neoplasms: case report and a comprehensive review of the literature. Am J Clin Oncol. 2003; 26: 79-83.
- Bagri PK, Singh D, Singhal MK, Guman Singh, Gaurav Mathur, Shankar Lal Jakhar, et al. Double Primary Malignancies: A Clinical & Pathological Analysis Report from a Regional Cancer Institute in India. Iran J Cancer Prev. 2014; 7: 66-72.
- Papaconstantinou I, Mantzos DS, Asimakoula K. A 12-year experience at a tertiary hospital on patients with multiple primary malignant neoplasms. J BUON. 2015; 2: 332-337.
- Hayat MJ, Howlander N, Reichman ME, Edwards BK. Cancer Statistics, Trends, and Multiple Primary Cancer Analyses from the Surveillance, Epidemiology and End Results (SEER) Program. The Oncologist. 2007; 12: 20-37.
- De Pangher Manzini V, Calucci F, Terpin MM, Loru F, Brollo A, Ramani L, et al. Multiple primary malignant tumors in patients with hepatocellular carcinoma. A review of 29 patients. Tumori. 1996; 82: 245-248.
- Xu W, Liao W, Ge Penglei, Ren J, Xu H, Yang H, et al. Multiple Primary malignancies in Patients With Hepatocellular Carcinoma. Medicine. 2016; 95: e 3491.
- Imada J, Hoshino H, Nishimura D, Morita K, Yoshida N, Katada N, et al. Case Report: multiple cancers: hepatocellular carcinoma and adenocarcinomas of the common bile duct and the gall-bladder in a woman with primary biliary cirrhosis. J Gastroenterol Hepatolo. 1996; 11: 546-550.
- Kim JW, Han JW, Jung SY, Jae Pil Jung, Jeong Won Kim. Synchronous double primary malignant tumor of the gallbladder and liver: a case report. World J Surg Oncol. 2011; 9: 84-87.
- La Greca G, Barbagallo F, Rodolico M, Greco L, Randazzo V, Scuderi M, et al. Hepatocarcinoma, gallstone disease and gall-bladder carcinoma: a case report of a rare incidental association. Chira Ital. 2004; 56: 571-574.
- Duffy A, Capanu M, Abou-Alfa GK, Huitzil D, Jarnagin W, Fong Y, et al. Gallbladder cancer (GBC): 10-year experience at memorial Sloan-Kettering Cancer Centre (MSKCC). J Surg Oncol. 2008; 98: 485-489.
- Mizuno T, Ebata T, Yokoyama Y, Tsuyoshi Igami, Gen Sugawara, Junpei Yamaguchi, et al. Adjuvant gemcitabine monotherapy for resectable perihilar cholangiocarcinoma with lymph node involvement: a propensity score matching analysis. Surg Today. 2017; 47: 182-192.
- Kobayashi S, Tomokuni A, Gotoh K, Takahashi H, Akita H, Marubashi S, et al. A retrospective analysis of the clinical effects of neoadjuvant combination therapy with full-dose gemcitabine and radiation therapy in patients with biliary tract cancer. Eur J Surg Oncol. 2017; 43: 763-771.