Case Report
Alcoholic Binge and Nontraumatic Rhabdomyolysis: A Case Report
Ravi Bansal1* and Savita Bansal2
1Department of Nephrology, Pushpawati Singhania Research Institute, India
2Department of Pathology, Manav Rachna Dental College, India
*Corresponding author: Ravi Bansal, Department of Nephrology, Pushpawati Singhania Research Institute, Sheikh Sarai-II, New Delhi-17, India
Published: 03 Jul, 2017
Cite this article as: Bansal R, Bansal S. Alcoholic Binge
and Nontraumatic Rhabdomyolysis: A
Case Report. Ann Clin Case Rep. 2017;
2: 1389.
Abstract
Alcoholism is on the rise and it has widespread impact on the health of a person. Non traumatic rhabdomyolysis is one such effect of rhabdomyolysis. Though this condition is reversible, early diagnosis and meticulous management of fluid electrolyte imbalance is crucial to prevent morbidity and mortality in these patients. We present here a case of 58 year old non-diabetic non hypertensive male with history of alcohol binge followed by unconsciousness. He presented with confusing neurological symptoms and acute kidney injury requiring dialysis therapy. The diagnosis of rhabdomyolysis was delayed by a day but subsequently managed successfully. We discuss the management challenges and give an inside to alcohol related rhabdomyolysis.
Introduction
Alcoholism has widespread impact on all organs of the body. With reports of benefits of mild to moderate alcohol consumption, the harmful effects of alcohol consumption are somewhat downplayed [1,2]. Acute Kidney Injury (AKI) following alcohol binge has also been reported earlier [3]. The mechanism of AKI associated with alcohol binge is multifactorial. Here we present a successfully managed case of alcohol binge associated non-traumatic rhabdomyolysis with AKI to highlight the importance of this entity.
Case Presentation
A 58yr old non diabetic, non-hypertensive, obese male was brought to the emergency room
with complains of pain, numbness and decrease in power of left upper and lower limb. Patient
was a known case of alcohol dependence syndrome on treatment with benzodiazepines. He had an
alcohol binge the previous night, approximately 500 ml, and fell unconscious. When he regained
consciousness he was not able to stand. On examination he was conscious oriented and febrile. There
was mild swelling and tenderness of right thigh though there was no history of injury. Neurological
examination showed decrease in power of right upper limb and lower limb. Deep tendon reflexes
and planters were mute on left side. Other systemic examinations were normal. There was no history
to suggest systemic disease or any positive family history.
Initial investigations revealed blood urea 67 mg/dl and serum creatinine of 1.3 mg/dl. He had
metabolic acidosis with pH =7.19 and bicarbonate levels (HCO3) 14.3 mmol/l. ECG showed sinus
tachycardia. MRI brain was normal. He was admitted under neurology and received one dose of
intra muscular diclofenac. He was clinically dehydrated and was given fluid supplements. Over next
24 hours he had urine output of around 1 liter. He was put on tablet librium by the psychiatrist.
On day 2 reports showed deranged renal functions and elevated liver enzymes. Urine output
decreased to 300 ml on day 2. His weakness of left side of body improved slowly. Ultrasound of
the thigh did not show any deep vein thrombosis but there was soft tissue edema and fluid in intermuscular
plains. Right kidney was 12.5 cm and left kidney 11.8 cm in size. There was grade II fatty
liver. A diagnosis of acute renal failure, possibly ATN was kept. Patient was managed with fluid
resuscitation, diet plan and counselled for possible need for hemodialysis. His serum creatinine had
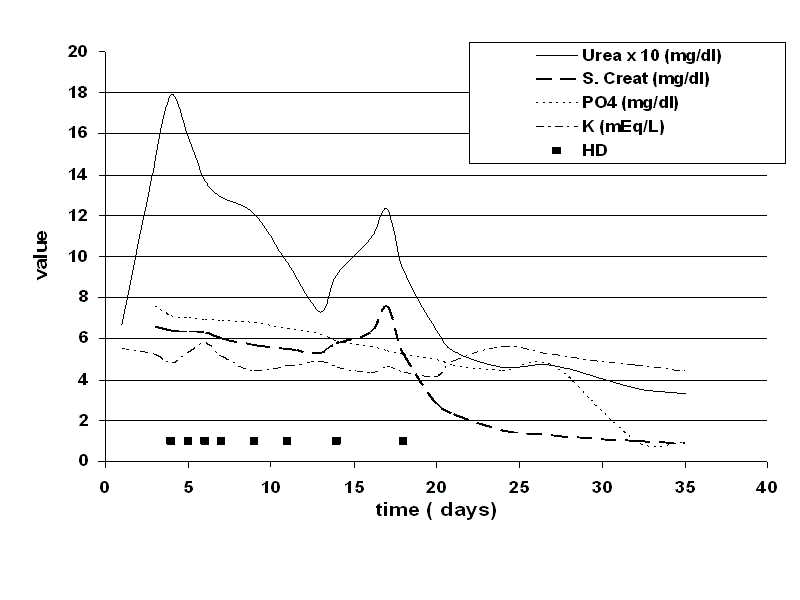
increased to 6.6 mg/dl, urine showed 2+ proteins, 5-7 RBC/HPF, serum CPK was 12100 IU/L and
Blood myoglobin level of 899 ng/ml (Table 1). A diagnosis of rhabdomyolysis with AKI was made. Patient was put on daily dialysis initially in view of rapidly rising creatinine and potassium levels.
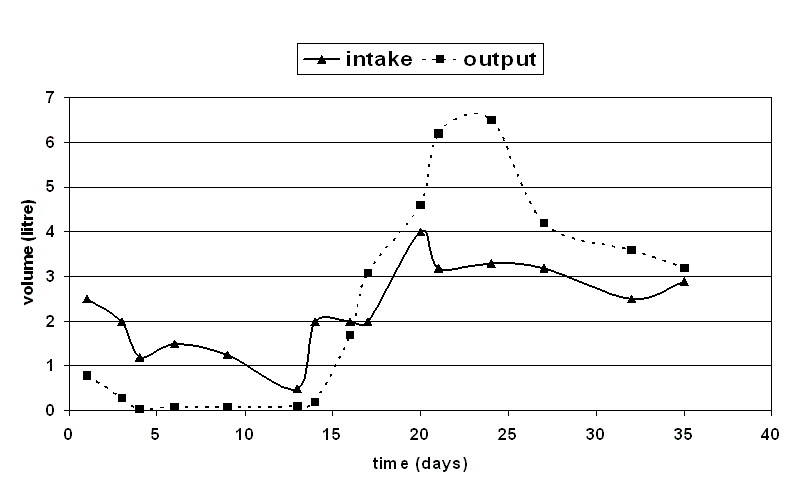
Patient remained almost anuric till 13 days of illness. Subsequently urine output started improving
and need for dialysis was reduced (Figure 1). Total he required 8 sessions of hemodialysis last on 18th
day of illness. Serum creatinine and urinary sediment completely normalized by 37th day of illness
(Figure 2).
For the assessment of Left upper and lower limb weakness patient
underwent MRI brain and spine which was essentially normal but
multiple nerve root compression in lumber spine secondary to
osteophytes were seen. His weakness started improving from 2nd day
and patient could walk with support from 20th day of illness. Nerve
conduction studies were suggestive of compressive nerve injury
and patient received physiotherapy for the same. Ultimately patient
recovered to full normal activity in next three months.
Table 1
Figure 1
Figure 2
Discussion
Our patient is a middle aged obese man, non-hypertensive non
diabetic with low risk of renal disease. He had an alcohol binge and lay
immobilized for few hours. This was followed by myoglobinuria and
swelling of left thigh, suggesting muscle injury. He was dehydrated on
presentation and had raised liver enzymes. He also received two doses
of NSAIDS. So he had all the possible factors to precipitate alcohol
binge associated AKI.
Most common concept of rhabdomyolysis is that of posttraumatic
muscle injury. First cases of crush syndrome and ARF reported
during the earthquake in Messina in 1908 and in World War I [4].
But in the modern times non-traumatic causes of rhabdomyolysis predominate [5]. These include occlusion or hypoperfusion of
vessels supplying the muscle during prolonged immobilization as
in lithotomy position, electrical current, hyperthermia, metabolic
myopathies, drugs and toxins (including alcohol), infections,
electrolyte abnormalities, endocrinal disorders and polymyositis [5].
Among drugs, rhabdomyolysis is most commonly seen with HMGCoA
reductase inhibitors; others include alcohol, antimalarials,
diuretics, fibrates and INH. The most recently widely used proton
pump inhibitors [6], levofloxacin [7], caffine [8], mefloquine [9],
pregabalin [10], and sildenafil [11] have also been regarded as risk
factors for rhabdomyolysis. In recent times there has been a fourfold
increase in number of patients hospitalized for exercise induced
rhabdomyolysis [12].
Myoglobin released from rhabdomyolysis is easily filtered at the
glomerulus and reabsorbed in the tubules. Dehydration and renal
vasoconstriction favor cast formation. Myoglobin can thus precipitate
in the glomerular filtrate, particularly in an acidic environment, finally
causing tubular occlusion and severe kidney damage [13]. But if urine
pH is increased greater than 6.5 only 4% of myoglobin precipitates [14]. Free iron released from myoglobin will initiate free radical
production and further enhances the ischemic damage. Percentage of
patients with rhabdomyolysis who develop acute kidney injury varies
from 13% to over 50% [15].
The adverse effects of ethanol on muscles have been experimentally
described several decades ago. Alcohol can induce rhabdomyolysis
by inhibiting calcium accumulation into the sarcoplasmic reticulum,
disrupting muscle cell membranes, and inhibiting the sodiumpotassium
ATPase pump which helps maintaining cellular integrity
[16]. Other causes may coexist in the same patient, so increasing the
likelihood of rhabdomyolysis. These include delirium tremens and/
or alcohol withdrawal seizures, muscle hypoxia due to prolonged
immobilization and limb compression, hypoperfusion following
volume depletion, and hypokalemia and hypophosphatemia
that frequently occurs in poorly nourished alcoholic patients
[17]. Myoglobin itself can contribute to vasoconstriction. Liver
dysfunction in alcohol binge produces systemic vasoactive factors,
which increase renal insufficiency [3]. Use of NSAIDS, which inhibit
cyclooxygenase, can contribute to AKI by interfering with regulation
of renal hemodynamics. Thus Alcohol intake has been associated
with renal failure even in the absence of any trauma.
There are many management issues in patients of rhabdomyolysis
associated AKI. Fluid accumulation in the affected limb contributes
to hypovolemia and renal failure. Our patient also presented
with dehydration and low blood pressure probably related to the
sequestered fluid in the injured muscle. Later in the disease course
if muscle recovers faster than the kidney, the fluid is redistributed
to the vascular compartment and can cause fluid overload in the
presence of renal failure. Initially hyperalbuminemia may occur due
to dehydration but latter malnutrition, inflammation and capillary
leak cause hypoalbuminemia as seen in our patient. S. Albumin
fell from 3.1 gm/dl to 2.6 gm/dl but again improved to 4.3 gm/dl at
full recovery. Patient may develop metabolic acidosis from organic
acids of muscle and lactacidosis. These contribute to decrease in
urinary pH and increase precipitation of myoglobin and urate in
the tubules. In early stages hypocalcaemia can occur due to calcium
accumulation and deposit in the injured muscle. Sometimes massive
calcification can occur [18]. This in combination with hyperkalemia
may cause arrhythmia or seizures [14]. Later this calcium is released
in the circulation and can cause significant hypercalcemia if calcium
supplements are being used. Similarly potassium, phosphate and uric
acid also accumulate.
Another important aspect is diagnostic markers for
rhabdomyolysis. Myoglobin is rapidly and unpredictably eliminated
from the blood by hepatic metabolism and filtration by the kidney.
Therefore tests for myoglobin in plasma and urine may not be
sensitive but it will cause dipstick positive for blood. Creatine kinase
levels > 5000 IU/l have been proposed as indication for treatment.
In our case we documented high myoglobin levels in blood and high
creatine phosphokinase (CPK) levels. Recent studies have shown that
high plasma concentration of myoglobin may correlate with increased
chance of AKI [19]. Treatment includes removing any precipitating
factors if identified. Hydration and alkalization of urine can reduce
precipitation of heme pigment. Supportive treatment with fluids to
maintain intravascular volume and frequent dialysis to adequately
control potassium, uremic toxins, phosphate, and acidosis is very
important in these highly catabolic patients. Special attention should
be given to nutrition as these patients can develop malnourishment.
Overall, these patients have good prognosis if timely diagnosed and
adequately managed.
The diagnosis is not difficult if the condition is kept in mind.
Definitely the incidence of rhabdomyolysis and myoglobinuria must
be higher as compared to what we detect since not all patients will
develop advanced renal failure and the diagnosis may be missed if
not investigated early. How common it is in alcoholics is hard to say
but with more and more people indulging in alcohol consumption
more cases are likely to be seen. Alcoholics are also prone to
prolonged immobilization which increases their risk of developing
rhabdomyolysis.
In conclusion we presented a case of alcohol binge associated
rhabdomyolysis with acute renal failure. We want to highlight the
need for high index of clinical suspicion for early diagnosis of the
condition as it can mimic other diagnosis. All effort should be made
to identify the triggering condition. Prompt management giving
special consideration to fluid and electrolyte balance can make the
difference between life and death to these patients.
References
- Mukamal KJ, Conigrave KM, Mittleman MA, Carlos A Camargo, Meir J Stampfer, Walter C Willett, et al. Roles of drinking pattern and type of alcohol consumed in coronary heart disease in men. N Engl J Med. 2003; 348: 109-118.
- Lieber CS. Medical disorders of alcoholism. N Engl J Med. 1995; 333: 1058-1065.
- Tsuboi N, Yoshida H, Shibamura K, Hikita M, Tomonari H, Kuriyama S, et al. Acute renal failure after binge drinking of alcohol and nonsteroidal antiinflammatory drug ingestion. Intern Med. 1997; 36: 102-106.
- Bywaters EGL, Beall D. Crush injuries with impairment of renal function. Br Med J. 1941; 1: 427-432.
- Vanholder R, Sever MS, Erek E, Lameire N. Rhabdomyolysis. J Am Soc Nephrol. 2000; 11: 1553-61.
- S J Duncan, C V Howden. Proton pump inhibitors and risk of rhabdomyolysis. Drug Saf. 2017; 40: 61-64.
- J Febin, R Oluronbi, CS Pitchumoni. Levofloxacin-induced rhabdomyolysis: a case report. J Med Case Rep. 2016; 10: 235.
- C Campana, PL Griffin, EL Simon. Caffeine overdose resulting in severe rhabdomyolysis and acute renal failure. Am J Emerg Med. 2014; 32: 111.e3–32:111.e4.
- I Comelli, G Lippi, A Magnacavallo, Cervellin G. Mefloquine-associated rhabdomyolysis. Am J Emerg Med. 2016; 34: 2250.e5–2250.e6.
- R Gunathilake, LE Boyce, AT Knight. Pregabalin-associated rhabdomyolysis. Med J Aust. 2013; 199: 624-625.
- DJ Oh. Sildenafil overdose can cause rhabdomyolysis and subjective visual perception changes. Nephrology. 2014; 19: 258.
- Christian Aalborg, Cecilie Rød-Larsen, Ingjerd Leiro, Willy Aasebø. Økning i antall pasienter med treningsindusert rabdomyolyse?. Tidsskr Nor Legeforen. 2016; 136: 1532-1536.
- UB Hendgen-Cotta, U Flögel, M Kelm, Rassaf T. Unmasking the Janus face of myoglobin in health and disease. J Exp Biol. 2010; 213: 2734-2740.
- X Bosch, E Poch, J Grau. Rhabdomyolysis and acute kidney injury. N Engl J Med. 2009; 361: 62–72.
- Cervellin G, Comelli I, Benatti M, Sanchis-Gomar F, Bassi A, Lippi G. Non-traumatic rhabdomyolysis: Background, laboratory features, and acute clinical management. Clin Biochem. 2017; S0009-9120: 30071.
- SK Song, E Rubin. Ethanol produces muscle damage in human volunteers. Science. 1972; 175: 327–328.
- JP Knochel. Neuromuscular manifestations of electrolyte disorders. Am J Med. 1990; 19: 1137-1143.
- Lopez JR, Rojas B, Gonzalez MA, Terzic A. Myoplasmic Ca2+ concentration during exertional rhabdomyolysis. Lancet. 1995; 345: 424-425.
- CY Chen, YR Lin, LL Zhao, Wen-ChiehYang, Yu-JunChang, Han-PingWu. Clinical factors in predicting acute renal failure caused by rhabdomyolysis in the ED. Am J Emerg Med. 2013; 31: 1062-1066.