Case Report
Senna Induced Blistering Diaper Dermatitis
Gregory Schweitz1, Dan Quan1,2* and Thomas Bostwick1,2
1Department of Emergency Medicine, Maricopa Integrated Health System, Phoenix, Arizona, USA
2Department of Emergency Medicine, University of Arizona College of Medicine, Phoenix, Arizona, USA
*Corresponding author: Dan Quan, Department of Emergency Medicine, University of Arizona College of Medicine, Phoenix, Arizona, USA
Published: 04 May, 2017
Cite this article as: Schweitz G, Quan D, Bostwick T. Senna Induced Blistering Diaper Dermatitis. Ann Clin Case Rep. 2017; 2: 1349.
Abstract
There is a plethora of potential diagnoses for diaper dermatitis. When abuse is a significant concern, elucidating the etiology is of the utmost importance. While the clinician should always consider abuse in the differential, an appreciation for the imitators of abuse is also essential.A16 month-old female presented to the emergency department after the development of diaper dermatitis. Further investigation revealed she ingested chocolate laxatives containing senna, later determined to be the etiology of etiology of the diaper lesion. Awareness of these less common presentations can facilitate more straightforward clinical assessment, easier disposition and guard the parents of these patients from the scrutiny they are often subjected to when abuse is a serious consideration.
Introduction
In diaper wearing pediatric patients, the differential diagnosis of a skin rash in the distribution of the perineum is wide and classically includes irritant dermatitis, allergic dermatitis, yeast infection, bacterial infection, atopic dermatitis, psoriasis, seborrhea and scabies among others. An additional consideration is child abuse. Differentiating infection or irritant diaper rash from abuse may be clinically challenging.
Case Presentation
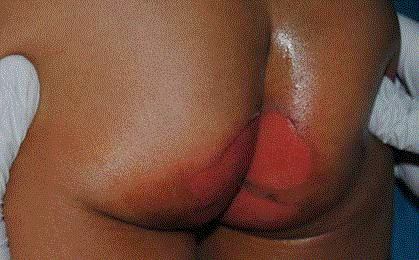
An otherwise healthy 16 month-old female toddler presented to the emergency department after escaping her playpen and accidentally ingesting three to four 15 mg chocolate laxatives containing senna. Per the patient’s mother the ingestion occurred two hours prior to arrival. One hour after ingestion she began having frequent, large, non-bloody soft stools in her diaper. These diapers were promptly changed and the patient was washed off in the shower under cold water. This was followed by the rapid development of a bright red, blistering wound with multiple clear bullae over the buttocks and perineum covering approximately 3% of total body surface area. No mucosal involvement was appreciated. The patient’s vital signs, review of systems, remainder of physical exam, allergy and medical history were unremarkable. The patient’s complete blood count, complete metabolic profile and urinalysis were within normal limits with only a mild elevation of segmented neutrophils to 68%. The urine drug screen, acetaminophen and salicylate levels were also negative. Wound culture grew occasional gram negative rods and gram positive cocci determined to be normal skin flora. There was some initial concern for scalded skin syndrome ergo the patient was started on cefazolin and clindamycin and admitted to the pediatric intensive care unit. After further evaluation this was considered highly unlikely and antibiotics were discontinued. The wounds were debrided and silver sulfadiazine was placed on affected areas. By the second day of admission, the patient’s bullae had all ruptured. Topical therapy was transitioned from silvadene to bacitracin and the patient was discharged that afternoon. During hospitalization, the patient’s vitals remained normal with only a brief maximum temperature of 38.1OC. She had no dietary difficulty and her stools returned to normal. The patient was seen at follow up visit 1 week later with near complete resolution of the wounds.
Discussion
In 1999 the US Food and Drug Administration removed phenolphthalein-based over-thecounter
laxatives from the market. The majority of these medications were replaced with senna on
the suspicion the former is carcinogenic [1,2]. Senna is derived from S. Alexandrina. Its side effect
profile is documented fairly well in the literature. These adverse effects range from theoretical injury
to the mesenteric nerve plexus, decreased skin immunity [3,4], asthma and rhinoconjunctivitis
[5,6] to near fatal hemorrhage in patients on warfarin [7]. Additionally, some suggest that senna is genotoxic however this has not been supported by current studies [8-10] and at least one indicates
that senna may have antioxidant properties [11].
Similar cases as above are described in one report citing 6 patients
with senna-induced-stool associated perianal dermatitis [12] as well
as a prospective observation study utilizing the databases of 6 poison
centers [13] examining these occurrences over the 9-month period.
One other case report presented a patient that developed a rash after
chronic senna use, however this case differed in that the rash was of
the non-blistering variety and the rash did not occur in an epidermal
region that came in contact with senna-induced stool. This incident
was presumed to be caused by senna-induced cytokine activation
in peripheral mononuclear cells in a susceptible patient [14] via
hematological and not topical transference.
While morbidity is fairly brief in these cases and there have been
no reported long-term sequelae or mortality, the clinician’s ability
to differentiate these skin wounds from those that occur with abuse
is paramount to the patient’s well being. In those cases, medical
providers must be suspicious. Since these findings may be highly
indicative of an unsafe environment and imply further emotional and
physical harm to the pediatric patient that may very well escalate at
the patient’s next presentation.
Skin wounds caused by chemical and/or thermal burns may
appear identical to the senna induced skin wound. The most salient
difference is in shape. The burn caused by abuse may be more
generalized and spread unevenly throughout the perineum and other
regions whereas the senna rash often is most intense at the perirectal
area and fades as it moves away from the source. This creates what has
been described as a diamond-shape very specific to the distribution of
the diaper indicating prolonged exposure to stool.
Figure 1
Conclusion
As in all cases of abuse, history is critical to elucidating the cause of these lesions. In the case above, the history relayed by multiple members of the household corroborated one another and the family appeared appropriately concerned about the patient. This in conjunction with a literature search at the time of presentation of senna-induced wounds contributed to the seemingly appropriate diagnosis being made.
References
- Hietala P, Marvola M, Parviainen T, Lainonen H. Laxative potency and acute toxicity of some anthraquinone derivatives, senna extracts and fractions of senna extracts. Pharmacol Toxicol. 1987;61(2):153-6.
- Laxative drug products for over-the-counter human use. Food and Drug Administration, HHS. Final rule. Fed Regist. 1999;64(19):4535-40.
- Yamate Y, Hiramoto K, Yokoyama S, Ooi K. Immunological changes in the intestines and skin after senna administration. Pharm Biol. 2015;53(6):913–20.
- Hennebelle T, Weniger B, Joseph H, Sahpaz S, Bailleul F. Senna alata. Fitoterapia. 2009;80(7):385-93.
- Helin T, Mäkinen-Kiljunen S. Occupational asthma and rhinoconjunctivitis caused by senna. Allergy. 1996;51(3):181-4.
- Marks GB, Salome CM, Woolcock AJ. Asthma and allergy associated with occupational exposure to ispaghula and senna products in a pharmaceutical work force. Am Rev Respir Dis. 1991;144(5):1065–9.
- Kittisupamongkol W, Nilaratanakul V, Kulwichit W. Near-fatal bleeding, senna, and the opposite of lettuce. Lancet. 2008;371(9614):784.
- Mitchell JM, Mengs U, McPherson S, Zijlstra J, Dettmar P, Gregson R, et al. An oral carcinogenicity and toxicity study of senna (Tinnevelly senna fruits) in the rat. Arch Toxicol. 2006;80(1):34-44.
- Mengs U, Mitchell J, McPherson S, Gregson R, Tigner J. A 13-week oral toxicity study of senna in the rat with an 8-week recovery period. Arch Toxicol. 2004;78(5):269-75.
- Heidemann A, Miltenburger HG, Mengs U. The genotoxicity status of senna. Pharmacology. 1993;47 Suppl 1:178-86.
- Silva CR, Monteiro MR, Rocha HM, Ribeiro AF, Caldeira-de-Araujo A, Leitão AC et al. Assessment of antimutagenic and genotoxic potential of senna (Cassia angustifoliaVahl.) aqueous extract using in vitro assays. ToxicolIn Vitro. 2008;22(1):212–8.
- Smith WA, Taintor AR, Kos L, Drolet B. Senna-Containing Laxative Inducing Blistering Dermatitis in Toddlers. Arch Derm. 2012; 148(3):402–3.
- Spiller HA, Winter ML, Weber JA, Krenzelok EP, Anderson DL, Ryan ML. Skin breakdown and blisters from senna-containing laxatives in young children. Ann Pharmacother. 2003;37(5):636–9.
- Norisugi O, Yoshihisa Y, Shimizu K, Shimizu T. In vitro cytokine expression by peripheral mononuclear cells in herbal drug-induced skin eruption. Acta Derm Venereol. 2014;94(1):58-62.