Case Report
Treatment Recommendations of a Rare Neurological Paraneoplastic Syndrome Caused by Small Cell Lung Cancer: A Case Report
Wenxing Yan, Daoming Zhang, Wanze Zhang, Min Liu and Linlin Liu*
Radiotherapy Department of Oncology, Second Hospital of Jilin University, Changchun 130021, People’s Republic
of China
*Corresponding author: Linlin Liu, Department of Radiotherapy, Second Hospital of Jilin University, Changchun 130021, People’s Republic of China
Published: 01 Mar, 2017
Cite this article as: Yan W, Zhang D, Zhang W, Liu M, Liu
L. Treatment Recommendations of
a Rare Neurological Paraneoplastic
Syndrome Caused by Small Cell Lung
Cancer: A Case Report. Ann Clin Case
Rep. 2017; 2: 1283.
Abstract
Paraneoplastic syndrome refers to these pathological changes of far organ function of patients
when malignancy do not metastasize. It would cause some relevant clinical manifestations in nerve,
digestion, hematopoiesis, endocrine, bones, joints and urinary system. And when the lesions occur
in nervous system, called the neurological Paraneoplastic Syndrome (PNS).
PNS occurs with any type of malignancy although the more commonly tumors are Small-Cell Lung
Cancer (SCLC). It appears generally earlier comparing with the clinical symptoms directly caused
by the tumor. The diagnosis is difficult and the treatment is more difficult. Neurological dysfunction
is severe.
This paper reports a case of 59 year old male with PNS as the initial symptom of small cell lung
cancer. The symptoms of PNS repeated attacks after surgery and chemotherapy, but the symptoms
are serious, it is rare in clinical.
After the multidisciplinary consultation, Symptoms of PNS in patients accepting the active treatment
of the original disease and anti-depressant drugs and anti-pain drug treatment at the same time have
eased, which has a certain reference value to the treatment of PNS.
Keywords: Small cell lung cancer (SCLC); Paraneoplastic neurological syndromes (PNS); MDT (Multi-disciplinary team); Treatment
Introduction
Currently the main treatments for PNS are anti-tumor therapy and immune therapy. The most effective means firstly considered should be actively looking for and therapy of primary tumor to clear the corresponding antigen. But after resection or chemoradiotherapy of the primary tumor, the symptoms of PNS have not eased much more. And immunomodulatory therapy has only curative effect to the PNS symptoms involving of the nerve-muscle or joint of peripheral nerve, but these caused by the CNS (Central Nervous System) is limited [1]. Despite the majority of patients have accepted the aggressive treatment, the results is unsatisfactory. In addition due to irreversible injury of Central neuron caused by the lack of neural function leads to the damage of lasting legacy. So the treatment of PNS has always been not satisfactory. This case shared us a successful case about the optimal treatment experience of PNS, hoping to have certain reference for clinicians, which also emphasized the importance of MDT at the same time.
Case Presentation
A 59-year-old male with no particular past medical history, was presented with the numbness
symptoms of face and limbs and misdiagnosed as peripheral neuritis. The numbness symptoms
were persisting after symptomatic treatment. The chest CT on June 26, 2015 revealed nodular
density in left lower lobe.
The patient accepted surgical treatment On October 14, 2015. Postoperative pathology: (the left
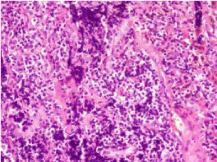
lower lobe) small cell carcinoma (Figure 1). Immunohistochemical staining: Syn (+), CgA (+), CD56
(+), TTF-1(+), Ki67 (+90%), LCA (-), P40 (-), CK5/6 (-), CK (AE1/AE3) (+) (Figure 2). Antibody
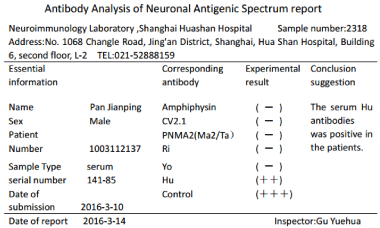
analysis of neuronal antigenic spectrum: Hu antibodies (++) (Figure 3). pT1N0M0. The numbness
symptoms of whole body had eased, but then continues to increase after postoperative 3 days. The patient received two cycle chemotherapy (etoposide + losplatinum)
and immune modulation at the same times between October 2015
to December 2015. The whole body numbness symptoms of the
patient would ease for 2-3 days after each treatment but it would
continue to aggravate. The patient received radiation therapy On
November 20, 2015. Radiation therapy was stopped because of
bone marrow suppression. The numbness symptoms of the patient
still persisted and even wanted to suicide. Later the patient received
Pregabalin, Bulleyaconitine A, Amitriptyline and Duloxetine after
Multidisciplinary consultation. PNS symptoms of the patient had
eased after Oral medication. The symptoms of PNS had completely
eased, the patient still be alive healthily by follow-up On December
2016.
Figure 1
Figure 2
Figure 3
Discussion
The pathogenesis of Neurological paraneoplastic syndrome,
also known as the remote effect of tumor is complex [2]. It could be
that antigen of the nervous system expressed in tumor cells occurs
immune cross-reactivity with nerve tissue, which cause nervous
system dysfunction. Another possible mechanism is that immune
responses occur when the apoptotic cells from the rapid growth and
apoptosis of tumor are engulfed by antigen-presenting cells, or tumor
cells from abnormal neural differentiation express nerve antigen,
which can trigger a series of immune reaction.
Hu antibody in diagnosis of small cell lung cancer with
paraneoplastic syndrome is highly sensitivity and specificity. And
Sentis Madrid H etc [3] think: Paraneoplastic syndrome could be
clearly diagnosed when the classic clinical syndrome combined with
Hu antibodies (+) in patients. Hu antibody of this patient is positive.
The numbness symptoms of face and limbs was seriously. The patients
had suicidal depression. So the diagnosis was clearly.
Now there is no randomised control trial data for the treatment
for PNS. But the main treatments For PNS include: (1) anti-cancer
treatments of surgery, radiotherapy, chemotherapy to control disease
progression; (2) the immunosuppressive therapy, such as hormones,
cyclophosphamide, plasma exchange and the treatment of the humoral immune method [4]. But for most patients, the effect of
immune therapy is poor and the main treatment is still controlling
the primary tumor.
In this case, we organized a multidisciplinary consultation and
decided that adjuvant therapy such as Pregabalin, Bulleyaconitine A,
amitriptyline, duloxetine were added on the basis of actively treating
the primary disease. The numbness symptoms of face and limbs had
eased after one month. Studies had shown that Pregabalin by reducing
excitatory neurotransmitter release such as glutamate, noradrenaline,
substance P, could inhibite neurons euphoria and treat spontaneous
pain after nerve injury, so as to alleviate neuropathic pain and allergic
symptoms [5-8]. Bulleyaconitine A, modern plant medicine, has a
good analgesics and anti-inflammatory. It also has certain immune
regulation. It has a certain analgesic effect by effectively reducing
pain relying on electro-physiological basis-sodiumion current [9].
The mechanism of duloxetine and amitriptyline is similar. They
can enhance the norepinephrine and serotonin to neurotransmitter
systems of the dual role of the nerve conduction, which is useful
for the regulation of emotion and pain sensitivity and can improve
tolerance of the body to pain [10-11]. These drugs have achieved a
certain effect to peripheral neuropathy induced by chemotherapy
[12-13].
The PNS symptoms of the patient were eased. The mechanism
may be that the secretion of abnormal hormone of tumor cells and
specific antigen of the nerve reduce, which weakens the ability of
the body's immune response of neural tissue. Therefore the whole
body numbness symptoms were alleviated, avoiding the emergence
of permanent neurological damage, improving the quality of life and
prolonging survival.
Conclusion
The incidence of Neurological paraneoplastic syndrome in patients with tumor is low. Conventional treatment effect is not satisfactory and poor prognosis. The numbness symptoms of the patient accepting Pregabalin and antidepressant drugs on the basis of the primary disease were eased, which provides an optimal reference treatment of neurological paraneoplastic syndrome.
Consent
Written informed consent was obtained from the patient for publication of this Case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Authors' Contributions
WY and DZ conceived the idea, did the literature search and prepared the manuscript. LL,WZ and ML provided critical review of the manuscript and research guidance. All authors have read and approved the final manuscript.
References
- Vedeler CA, Antoine JC, Giometto B, Graus F, Grisold W, Hart IK, et al. Management paraneoplastic neurological syndromes: report of an EFNS Task Force. Eur J Neurol 2006; 13: 682-690.
- Hollis A. Paraneoplastic syndromes. Equine Vet Educ. 2011; 23: 184-185.
- Senties-Madrid H, Vega-Boada F. Paraneoplastic syndromes associated with anti-Hu antibodys. Isr Med Assoc J. 2001; 3: 94-103.
- Giometto B, Vitaliani R, Lindeck-Pozza, Grisold W, Vedeler C. Treatment for paraneoplastic neuropathies. Cochrane Database Syst Rev. 2012; 12: CD007625.
- Sills GJ. The mechanisms of action of gabapentin and pregabalin. Curr Opin Pharmacol. 2006; 6: 108-113.
- Taylor CP, Angelotti T, Fauman E. Pharmacology and mechanism of action of pregabalin: the calcium channel alpha2-delta (alpha2-delta) subunit as a target for antiepileptic drug discovery. Epilepsy Res. 2007; 73: 137-150.
- Tzellos TG, Papazisis G, Toulis KA, Sardeli Ch, Kouvelas D. A2-delta ligands gabapentin and pregabalin: future implications in daily clinical practice. Hippokratia. 2010; 14: 71-75.
- De Santis S, Borghesi C, Ricciardi S, Giovannoni D, Fulvi A, Migliorino MR, et al. Analgesic effectiveness and tolerability of oral oxycodone/ naloxone and pregabalin in patients with lung cancer and neuropathic pain: an observational analysis. Onco Targets and Therapy. 2016; 9: 4043– 4052.
- Wang CF, Gerner P, Wang SY, Wang GK. Bulleyaconitine A isolated from Aconitum plant displays long-acting local anesthetic properties in vitro and in vivo. Anesthesiology. 2007; 107: 82-90.
- Detke MJ, Lu Y, Goldstein DJ, Hayes JR, Demitrack MA. Duloxetine, 60 mg once daily, for major depressive disorder: a randomized double-blind placebo-controlled trial. J Clin Psychiatry. 2002; 63: 308-315.
- Gupta S, Nihalani N, Masand P. Duloxetine: review of its pharmacology, and therapeutic use in depression and other psychiatric disorders. Ann Clin Psychiatry. 2007; 19: 125-132.
- Saif MW, Syrigos K, Kaley K, Isufi I. Role of pregabalin in treatment of oxaliplatin-induced sensory neuropathy. Anticancer Res. 2010; 30: 2927- 2933.
- Kaley TJ, Deangelis LM. Therapy of chemotherapy-induced peripheral neuropathy. Br J Haematol. 2009; 145: 3-14.