Case Report
Mixed Oncocytoma and Papillary Renal Cell Carcinoma
Dilek E. Baydar*
Department of Pathology, Hacettepe University School of Medicine, Turkey
*Corresponding author: Dilek Ertoy Baydar, Department of Pathology, Hacettepe University Hospital, Department of Pathology, Sihhiye / Ankara, 06100 Turkey
Published: 23 Jun, 2016
Cite this article as: Baydar DE. Mixed Oncocytoma and
Papillary Renal Cell Carcinoma. Ann
Clin Case Rep. 2016; 1: 1025.
Abstract
The hybrid tumors composed of oncocytoma and chromophobe renal cell carcinoma (ChRCC) are known to occur. They are seen in 3 settings, namely Birt-Hogg-Dubé Syndrome, renal oncocytosis, and as sporadic neoplasia. However, mixed renal tumors composed of a component other than ChRCC in addition to oncocytoma are extremely rare. Herein, a renal cell neoplasm consisted of two intermingled components, namely oncocytoma and papillary renal cell carcinoma were presented.
Introduction
Renal oncocytomas comprise 3–7% of all renal tumors, and are biologically benign [1,2]. They
consist of uniform and round or polygonal cells that contain abundant eosinophilic granules in the
cytoplasm that have been identified as mitochondria electron microscopically [3]. Neoplastic nests
usually lie in a loose edematous stroma. The cell of origin of renal oncocytoma (RO) is thought to be
the intercalated cells of collecting tubules [4].
Papillary renal cell carcinoma (PRCC) is the second most common subtype of renal cell
carcinoma. It is believed to derive from proximal tubular epithelium [5]. This tumor exhibits
papillary or tubulopapillary architecture. PRCC has been conventionally divided into two types.
In type I carcinomas, papillae are lined by single layer of cells often with scanty pale cytoplasm.
Type 2 PRCCs show cells with higher nucleolar grade and abundant eosinophilic cytoplasm, and
pseudostratification.
Coexistence of PRCC and RO within the same tumoral mass is extremely unusual. There are
only 7 such cases previously documented in the literature up to now. In each of these reports, an
oncocytoma has been the main tumor and a smaller focus of papillary renal cell neoplasia has been
found embedded within it. In this article, a unique case of intimately mixed RO and PRCC in a
single tumor mass is presented.
Case Report
An 49-year-old man was referred to our hospital after detection of a renal mass on abdominal
ultrasonography during regular medical check-up. His physical examination was unremarkable. His
urinalysis and other routine blood tests were normal. Computed tomography disclosed that the mass
was located at the upper pole of right kidney, was 4.2×4.0×3.4 cm in size and well circumscribed
with contrast enhancement. Right partial nephrectomy was carried out.
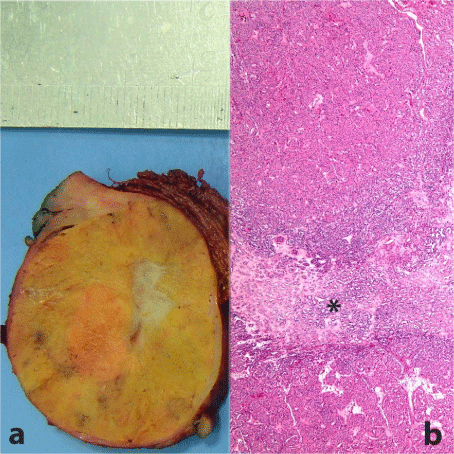
Macroscopical examination revealed 4.5x4x3.5 cm well-circumscribed solid lesion in the
renal parenchyma, yellowish/tan in color with white stellate scar in the center (Figure 1A). Light
microscopy showed that the tumor was composed of two distinct elements differing both in pattern
and cell morphology (Figure 1B). The first component contained nests of larger cells with dark
eosinophilic cytoplasm and rounded regular nuclei, reminiscent of oncocytoma. Within this, closely
blended with oncocytic nests, there was a second component made up of tubules and papillae.
Tubulopapillary structures were formed by cells with different cytologic features than oncocytoma
cells; they had narrower cytoplasm and oval small nuclei.
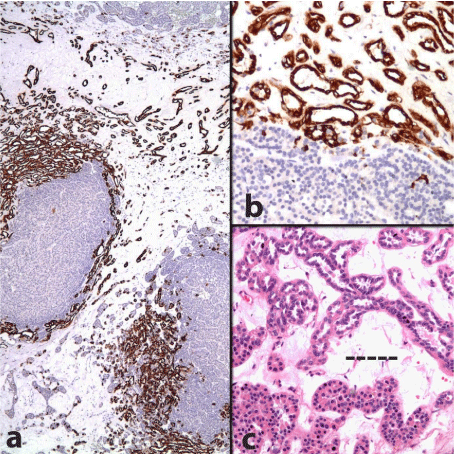
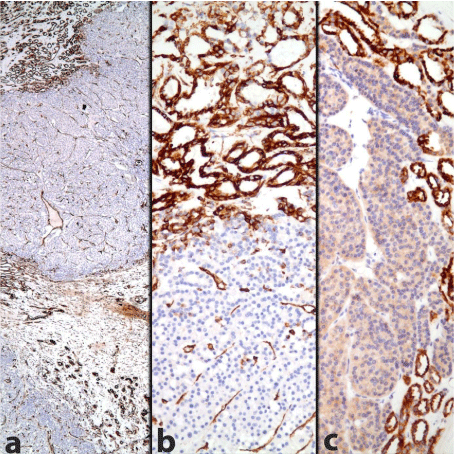
Immunohistochemical stainings demonstrated strong expression of cytokeratin 7, vimentin,
α-methylacyl-CoA racemase and CD10 in the tubulopapillary areas (Figure 2 and 3). Oncoytoma
component was negative for these markers but showed positivity for CD117. Based on its
morphological features and immune profile, the tumor was diagnosed as mixed renal oncocytoma
and type I PRCC.
There was no extra capsular tumor infiltration into perinephric fat tissue. Neither necrosis nor
lymphovascular permeation was seen. Surgical margins were free of tumor.
Figure 1
Figure 1
a) Macroscopical picture of tumor showing well-defined spherical
solid mass, yellow in color and with stellate scar in the center. b) Intermixed
two distinct components microscopically: pale looking area in the middle
having morphology of papillary RCC (asterisk), and oncocytic upper and
lower regions (H-E x 40).
Figure 2
Figure 2
a) and b) Cytokeratin 7 highlighting papillary renal cell carcinoma
component. It is not expressed by oncocytoma part (a: Immunohistochemistry,
anti-cytokeratin 7 Ab x 40; b: Immunohistochemistry, anti-cytokeratin 7 Ab x
200). c) H-E stain with a dashed line separating papillary renal cell carcinoma
at the upper part from oncocytoma at the lower half (H-E x 200).
Figure 3
Figure 3
Immunohistochemistry documenting the presence of two different
constituents within the tumor: Vimentin and AMACR are positive in papillary
RCC, but negative in oncocytoma (Immunohistochemistry, a: anti-vimentin
Ab x 40, b: anti-vimentin Ab x 200, c: anti-AMACR Ab x 200).
Discussion
Renal oncocytoma (RO) is a neoplasm of renal epithelial
origin that has been thought to be derived from intercalated cells
of collecting tubules [4]. On the other hand, papillary renal cell
carcinoma (PRCC) is considered as proximal tubular origin [5]. ROs
occasionally coexist with chromophobe renal cell carcinomas that
similarly develop from intercalated cells [6]. However, coexistence of
RO with papillary renal cell neoplasm is extremely rare even though P
RCC is the second most frequent pathological subtype of RCC. There
are only 7 such cases formerly reported in the literature [7-13] (Table
1). The age of patients in the previous reports ranged between 60
and 78, our patient was the youngest among them being 49. Clinical
presentations were various, such as abdominal pain, normocytic
anemia, lethargy, anorexia and hypercalcemia as well as hematuria.
Three cases including ours were discovered incidentally. Total tumor
size varied between 1.5 cm and 6 cm in diameter, was 4.5x4x3.5 cm in
the current case. In all these lesions, the PRCC represented the smaller
component and showed low grade nuclei. Our case differed from the
others in that the PRCC within oncocytoma was not well delineated
focus, but rather two components were intermingled closely. This is
in contrast to former cases where PRCC was distinct on both gross
and microscopic examination within oncocytoma.
None of the tumors had evidence of tumor recurrence or
metastasis. This is consistent with low size and grade of PRCC
component. In fact, according to last WHO (2016) classification of
renal neoplasia, most previous lesions would be considered papillary
renal tubular adenomas today instead of papillary renal cell carcinoma
as the cut-off size between these two entities has been raised to 1,5cm
from 0.5 cm [14]. Only our case and the case reported by Özer et al.
[12] have papillary renal cell neoplasia above this threshold diameter
and can be diagnosed as PRCC today.
Renal oncocytoma has unique histologic features including
an organoid and tubulocystic architecture, myxoid or hyalinized
stroma, and occasionally some atypical findings including nuclear
pleomorphism, prominent nucleoli, and adjacent renal parenchymal
and perinephric fat involvement. By definition it lacks areas of clear
cell carcinoma or conspicuous papillary arrangement. The current
case demonstrating papillary formations imposes the differential
diagnosis of the lesion with PRCC showing oncocytic change [15].
PRCC with oncocytic cytoplasm and oncocytoma like low-grade
nuclei have been called oncocytic papillary renal cell carcinomas [16]
although they are not considered as a spesific subtype of RCC in the
last WHO (2016) renal neoplasia classification due to their incomplete
characterization yet. Our case differed from oncocytic papillary RCC
in that it had two distinct components both morphologically and
immunohistochemically. Areas of papillary RCC have demonstrated
strongly positive staining of CK7, CD10, vimentin and alphamethylacyl-CoA
racemase (AMACR), which is characteristic of
PRCC [17] whereas these markers have stained negative in oncocytic
regions. Papilla formations have been limited to papillary RCC areas
and not found in oncocytic regions. Furthermore, papillae have lining
cells with pale narrow -but not copious eosinophilic- cytoplasm.
The association between oncocytoma and papillary renal cell
neoplasia is hard to explain since there are only scarce previously
reported cases. Since they have different genetic backgrounds, it is
not possible to suggest common pathogenesis. Most likely, they are
collision tumors that happen to arise together at the same site by
coincidence. However their existence creates a concern thinking that
when pathology is about to make a call of oncocytoma in a tumor
biopsy sample, the possibility of unsampled malignant component
might be needed to be brought into the consideration. These tumors
pose challenges to the urologists, pathologists and oncologists who
would struggle in their classification and choosing right form of
patient management.
Oncocytomas are benign proliferations but papillary RCC has
malignant potential. Hybrid tumors containing both elements are
difficult to approach clinically. Novel cases with these types of rare
tumors are needed to be reported in the literature so that additional
information concerning optimal treatment policy and the prognosis
of these patients can be obtained.
References
- Amin MB, Crotty TB, Tickoo SK, Farrow GM. Renal oncocytoma: a reappraisal of morphologic features with clinicopathologic findings in 80 cases. Am J Surg Pathol. 1997; 21: 1-12.
- Trpkov K, Yilmaz A, Uzer D, Dishongh KM, Quick CM, Bismar TA, et al. Renal oncocytoma revisited: a clinicopathological study of 109 cases with emphasis on problematic diagnostic features. Histopathology. 2010; 57: 893-906.
- Eble JN, Hull MT. Morphologic features of renal oncocytoma: a light and electron microscopic study. Hum Pathol. 1984; 15: 1054-1061.
- Kuroda N, Toi M, Hiroi M, Shuin T, Enzan H. Review of renal oncocytoma with focus on clinical and pathobiological aspects. Histol Histopathol. 2003; 18: 935.
- Rini BI, Campbell SC, Escudier B. Renal cell carcinoma. Lancet. 2009; 373: 1119-1132.
- Fleming S. Distal nephron neoplasms. Semin Diagn Pathol. 2015; 32: 114-123.
- Al-Saleem T, Balsara BR, Liu Z, Feder M, Testa JR, Wu H, et al. Renal oncocytoma with loss of chromosomes Y and 1 evolving to papillary carcinoma in connection with gain of chromosome 7. Coincidence or progression? Cancer Genet Cytogenet. 2005; 163: 81-85.
- Vasuri F, Fellegara G. Collision renal tumors. Int J Surg Pathol. 2009; 17: 338-339.
- Rowsell C, Fleshner N, Marrano P, Squire J, Evans A. Papillary renal cell carcinoma within a renal oncocytoma: case report of an incidental finding of a tumour within a tumour. J Clin Pathol. 2007; 60: 426-428.
- Floyd MS, Javed S, Pradeep KE, De Bolla AR. Composite oncocytoma and papillary renal cell carcinoma of the kidney treated by partial nephrectomy: a case report. Scientific World Journal. 2011; 11: 1173-1177.
- Sejben I, Szabó Z, Lukács N, Loránd M, Sükösd F, Cserni G. Papillary renal cell carcinoma embedded in an oncocytoma: Case report of a rare combined tumour of the kidney. Can Urol Assoc J. 2013; 7: E513-516.
- Özer C, Gören MR, Egilmez T, Bal N. Papillary renal cell carcinoma within a renal oncocytoma: Case report of very rare coexistence. Can Urol Assoc J. 2014; 8: E928-930.
- Goyal R, Parwani AV, Gellert L, Hameed O, Giannico GA. A collision tumor of papillary renal cell carcinoma and oncocytoma: case report and literature review. Am J Clin Pathol. 2015; 144: 811-816.
- Eble J, Moch H, Amin MB. Papillary adenoma. In WHO Classification of Tumours of the Urinary System and Male Genital Organs. Moch H, Humphrey PA, Ulbright TM, Reuter VE, Editors. 4th ed. IARC, Lyon, 2016. p42.
- Munari E, Eccher A, Segala D, Iannucci A, Gobbo S, Chilosi M, et al. Oncocytic papillary renal cell carcinoma: potential pitfall in small enucleation. Pathologica. 2012; 104: 98-100.
- Lefèvre M, Couturier J, Sibony M, Bazille C, Boyer K, Callard P, et al. Adult papillary renal tumor with oncocytic cells: clinicopathologic, immunohistochemical, and cytogenetic features of 10 cases. Am J Surg Pathol. 2005; 29: 1576-1581.
- Truong LD, Shen SS. Immunohistochemical diagnosis of renal neoplasms. Arch Pathol Lab Med. 2011; 135: 92-109.