Case Report
A Case of a Cervical Cancer Patient with Elevated CEA Caused by Hashimoto’s Thyroiditis
Lei Gao1*, Gong Li1, Xiaojuan Li2, Cheng Xiao2 and Shucong Zhang2
1Department of Radiotherapy, Guangdong Provincial Hospital of Chinese Medicine, China
2Department of Medical Oncology, Second Clinical College of Guangzhou University of Chinese Medicine, China
*Corresponding author: Lei Gao, Radiotherapy Department, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, China
Published: 06 Oct, 2017
Cite this article as: Gao L, Li G, Li X, Xiao C, Zhang S. A
Case of a Cervical Cancer Patient with
Elevated CEA Caused by Hashimoto’s
Thyroiditis. Ann Clin Case Rep. 2017;
2: 1438.
Abstract
Carcinoembryonic Antigen (CEA) is a broad-spectrum tumor maker that can be expressed in a
variety of malignancies, it relatively high sensitivity, and can be used as an index for evaluation
therapeutic effect. Its level has been shown to be associated with tumor burden with colorectal
cancer. However, it is also elevated to a significant degree in non-malignant conditions. The purpose
of this case report is to describe Hashimoto’s thyroiditis causing a marked elevation of CEA levels
and to review other benign causes of elevated CEA.
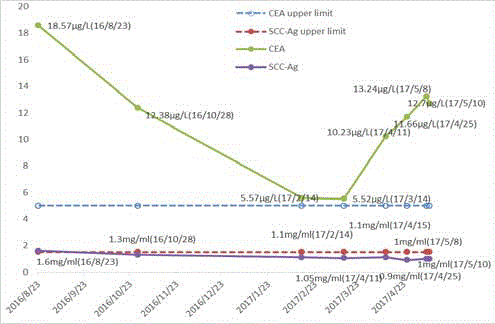
A 77-year-old woman was diagnosed with cervical cancer, and received radical chemoradiotherapy.
All data showed that the treatment was effectively, but repeated serum CEA levels were found an
upward trend, so we initially thought that was the result of tumor recurrence or other malignancies.
Then, we conducted further assessment, the SCC-Ag level was at the low state, it was an important
indicator of the effectiveness of cervical cancer treatment. And the rest of other tumor markers,
which including serum AFP, CA12-5, CA19-9, CA15-3, were all within the normal limit. On the
other hand, follow-up imaging examinations, including PET, CT and MRI, showed that treatment
was effectively, and there were no primary recurrence or metastasis. It is noteworthy that all
imaging studies suggest the sign of the thyroid gland was changed, but the patient did not have
overt hyperthyroid or hypothyroid features. With our further evaluation, serum thyroid peroxidase
antibody (aTPO), and thyroglobulin antibody titers (ATG) were markedly elevated. On thyroid
ultrasonography revealed the increase of the volume of thyroid gland, and multiple isoechoic
nodules, the patient was diagnosed with Hashimoto thyroiditis.
Keywords: Carcinoembryonic antigen; Hashimoto’s thyroiditis; Cervical Cancer
Introduction
Carcinoembryonic antigen (CEA) is an oncofetal glycoprotein, what normally expressed by
mucosal cells. It is over expressed in patients with adenocarcinoma like endometrial cancer colorectal
cancer, etc. And it is elevated to significant degree in variety of other malignancies, such as breast,
lung, stomach, and pancreas. And CEA is also expressed on the cell surface in certain inflammatory
conditions, including inflammatory bowel disease, peptic ulcer disease and hypothyroidism.
CEA is widely used as a screening tool for cancer. However, there are a number of nonmalignant
etiologies of elevated CEA level. Therefore, clinicians need to aware of it rare associations
so as to avoid unnecessary investigations without missing the malignant disorders. On the other
hand, CEA serum levels have been reported elevated in patients with autoimmune thyroid disease
[1]. We report the case of a patient with an elevated CEA level who underwent investigations for
cervical cancer, which was later found to be related to Hashimoto’s thyroiditis.
Case Presentation
A 77-year-old female with past medical history of hypertension, diabetes mellitus (DM),
nephrolithiasis and fatty livers. On August 18, 2016, she was diagnosed as cervical cancer;
pathological result was FTGO stage II b, squamous-cell carcinoma. Elevated serum CEA was
raised at 18.57μg/L (normal range 0~5μg/L), and squamous cell carcinoma antigen (SCC-Ag) was
1.6mg/ml (normal range 1.5mg/ml), which associated with squamous cell carcinoma (HCC) and
as an important indicator of the effectiveness or prognosis of cervical cancer treatment. The rest of
other tumor markers, which including serum AFP, CA12-5, CA19-9, CA15-3, were all within the
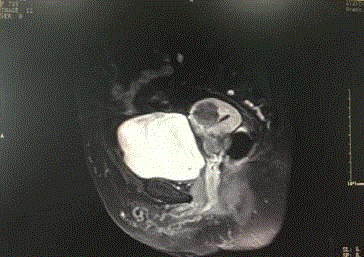
normal limit. At the same time, a MRI scan showed that the posterior wall of cervix was occupied, which accord with cervical cancer, and myoma of uterus? Therefore
she received radical radiotherapy. From September 19 to October
28, she had undergone external radiotherapy (ERBT), that a total
does of 45Gy/25Fx for five weeks. To investigate the short-term
clinical outcomes, on October 28, she was evaluated. Repeat serum
CEA was 12.38μg/L, which was lower than before. And compared
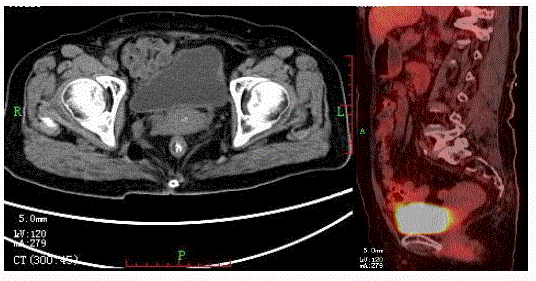
previous imaging results, a positron emission tomography (PET)
scan showed that treatment is effectively, because that the focus of
tumor was limited, and negative for hypermetabolic lesions, besides,
a sign of the thyroid gland was abnormal, considering nodular goiter.
However, as she did not have overt hyperthyroid or hypothyroid
features, treatment was not started. Subsequently she received a total
four times brachy therapy with dose of 6Gy/Fx, and plus three cycles
concurrent chemotherapy with oral capecitabine 1.0g bid, from
day1 to day4, as adjuvant therapy. Repeat serum CEA was noted to
decrease to 5.57μg/L, SCC-Ag was 1.3mg/ml, and MRI findings were
roughly the same as PET. Five months after radical treatment, the
patient tumor markers were demonstrated normal, and SCC-Ag level
was 1.1μg/L, expect for CEA that was increased to 10.23μg/L.
Initially, we considered whether there was recurrence or
metastasis. But follow-up CT scans were no evidence of primary
recurrence or metastasis, at the subsequent review, the CEA level
continued to rising, range from 11.6μg/L to 13.24μg/L, the level
of other tumor makers were within normal range, and all imaging
studies did not reveal any evidence of primary recurrence, metastasis
or other malignancy. There was one noteworthy, that all imaging
results suggest the sign of the thyroid gland had changed, so we
conducted further evaluation. On physical examination, we found
that the size of thyroid was observed to be increased and extended to sternocleidomastoid region; there were non-tender mass and no
prominent lymph nodes in the neck. Serum TSH was 0.441mIU/L
(normal level 0.5 to 4.5mIU/L), free T3 and free T4 were normal,
and serum thyroid peroxidase antibody (>1,300IU/ml; normal
range<60IU/ml; strongly positive), thyroglobulin antibody titers
(>500 IU/ml, normal range<60IU/ml) were markedly elevated. On
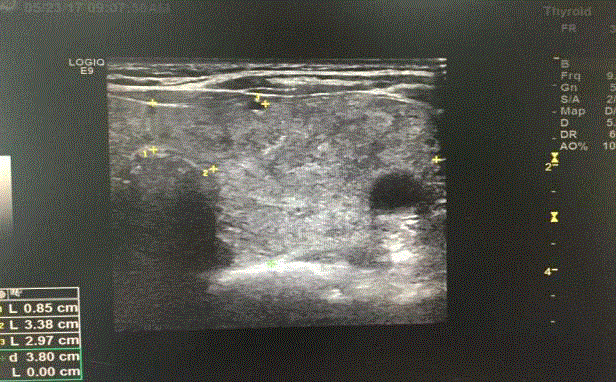
thyroid ultrasonography revealed the increase of the volume of
thyroid gland, and multiple isoechoic nodules, range from 4mm to
36mm. In the left lobe of the thyroid, the largest of which measured
34 mm × 36mm × 24mm, and the right lobe of thyroid gland was
measured a 14 mm × 14 mm × 9 mm-sized isohypoechioc nodule.
There were no prominent lymph nodes in the neck. All data was
consistent with Hashimoto’s thyroiditis (Figure 1 -4).
Figure 1
Figure 2
Figure 3
Figure 4
Discussion
In this report, we describe a case of a cancer patient with
evaluated level of CEA caused by Hashimoto’s thyroiditis. Although
the exact mechanism causing high serum CEA levels in patients
with Hashimoto’s thyroiditis remains unknown and would need
further studies to elucidate the pathophysiology. To our knowledge
and literature, this is not the first case report linking Hashimoto’s
thyroiditis with an elevated CEA level [2]. Keiji Inia et al. [1] reported
a case of myxedema coma caused by isolated thyrotropinstimulating
hormone deficiency and Hashimoto’s thyroiditis, that observed
possesses transient elevated levels of CEA, which rapidly decreased
after the hypothyroidism was treated.
The increase of CEA level could be caused by various factors,
such as, the presence of immunologically ross-reacting CEA-like
substances, or increased CEA synthesis, decrease CEA degradation.
In the current studies, these have shown hypothyroidism as potential
cause of the elevated CEA level, for example, Tukamu et al. [3]
observed that the mean CEA concentration was significantly higher in
hypothyroid patients than in normal and hyperthyroid patients, which means when patients are suffering from hypothyroidism, caution
should be taken when reading the tumor marker concentration. In
another study, found that increased CEA is not coming from the
thyroid itself. A significantly high frequency of positive CEA was
observed in hypothyroid patients with Hashimoto's disease, these
findings indicate that elevated CEA levels in hypothyroid patients do
not necessarily indicate malignancy [4].
Conclusion
Our case highlights the importance of awareness of the associations of elevated tumor markers with not just malignant conditions but also benign ones. The evaluation of prognosis of the tumor should be cautious with the interpretation of CEA. Whether to check the thyroid-related biochemical indexes, to exclude the relationship with Hashimoto’s thyroiditis, when cancer patients in the process of examination found CEA level continuous increased. Or patients with elevated CEA should also be evaluated for underlying thyroid disorders that may be sub-clinical.
References
- Keiji Iida, Yasuhisa Hino, Takeshi Ohara, Kazuo Chihara. A case of myxedema coma caused by isolated thyrotropin stimulating hormone deficiency and Hashimoto’s thyroiditis. Endocr J. 2011; 58: 143-148.
- Asad Ur Rahman F, Saif M W. Elevated Level of Serum Carcinoembryonic Antigen (CEA) and Search for a Malignancy: A Case Report. Cureus. 2016; 8: e648.
- Hashimoto T, Matsubara F. Changes in the Tumor Marker Concentration in Female Patients with Hyper-, Eu-, and Hypothyroidism. Endocrinol Jpn. 1989; 36: 873-879.
- Amino N, Kuro R, Yabu Y, Takai SI, Kawashima M, Morimoto S, et al. Elevated Levels of Circulating Carcinoembryonic Antigen in Hypothyroidism. Journal of Clinical Endocrinology and Metabolism. 1981; 52: 457-462.