Case Report
OSA Masquerading as Failure to Thrive and Developmental Delay: A Case Report
Katia El Taoum* and Supriya Jambhekar
Department of Pediatrics, Pulmonary and Sleep Division, University of Arkansas for Medical Sciences, Little Rock,
Arkansas
*Corresponding author: Katia El Taoum, Department of Pediatrics, Pulmonary and Sleep Division, Arkansas Children’s Hospital, 1 Children’s Way, Little Rock AR, 72202, Slot 512-17, 501-364-1006, USA
Published: 02 Oct, 2017
Cite this article as: El Taoum K, Jambhekar S. OSA
Masquerading as Failure to Thrive and
Developmental Delay: A Case Report.
Ann Clin Case Rep. 2017; 2: 1436.
Abstract
An 8-month-old girl was admitted to the hospital for failure to thrive with significant developmental delays. Snoring and hypoxia during sleep were noted and she was started on heated high-flow nasal cannula at 21% FiO2. She continued to have drops in her oxygen saturation down to 68%, in addition to retractions and tracheal tugging during sleep. Adenoidectomy was performed, removing 100% obstructing adenoids, but snoring and oxygen desaturations while sleeping continued. Polysomnography revealed severe obstructive sleep apnea (OSA) (apnea/hypopnea index (AHI) 160.8/hour, 236.6/hour in active sleep), and oxygen saturation nadir was 65%. Other surgeries were thought neither possible nor safe at that time. Auto titrating continuous positive airway pressure (autoCPAP) 4-6 cmH2O was started and later increased to 4-10 cmH2O. Oxygen saturation normalized on CPAP. Eleven days after initiation of treatment, the patient was able to hold her head up, reach for and play with toys, sit up and pull to stand with minimal assistance. She had a 1 kilogram weight gain during the hospital stay. By 14 months of age, she was standing with support and trying to speak. By 17 months, she had grown from below the 1st percentile to almost the 50th percentile for BMI. A tonsillectomy and uvulopalatopharyngoplasty were performed at that time resolving the OSA (AHI 0.4/hour, oxygen nadir 91% per PSG), and CPAP was discontinued. This case is interesting for pediatricians because it highlights the importance of having a high index of suspicion for OSA in children with failure to thrive or developmental delay, it points out the role of noninvasive ventilation for OSA treatment in infants, and It highlights the importance of follow up and continued re-evaluation for availability of surgical options in young children with OSA.
Abbreviations
ADHD: Attention Deficit Hyperactivity Disorder; AHI: Apnea Hypopnea Index; ALTE: Acute Life Threatening Event; Auto CPAP: Auto Titrating Continuous Positive Airway Pressure; CPAP: Continuous Positive Airway Pressure; FTT: Failure To Thrive; GERD: Gastro Esophageal Reflux Disease; NC: Nasal Cannula; NIV: Non Invasive Ventilation; OSA: Obstructive Sleep Apnea; PSG: Poly Somno Graphy
Table of Content Summary
FTT can result from different etiologies. This article shows an unusual cause of FTT and the challenges to diagnose it and treat it appropriately.
Contributors’ Statement Page
Dr El Taoum drafted the initial manuscript and Dr Jambhekar reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Introduction
Obstructive Sleep Apnea (OSA) is characterized by partial airway obstruction or intermittent
complete obstruction that interrupts normal ventilation during sleep [1]. Infants are at high risk of
severe OSA compared to older children and adults due to their upper airway anatomy, laryngeal
chemoreceptors, and sleep-related breathing patterns [2]. The most common presenting symptoms of OSA in infants are snoring, nocturnal hypoxia, and acute life threatening event (ALTE). In
children, OSA is known to be an important risk factor for development of behavioral dysfunction,
attention deficit hyperactivity disorder (ADHD), failure to thrive (FTT), and developmental delay if
not recognized and treated early [3].
FTT may result from multiple etiologies. Non organic etiologies
are more frequently encountered, but pediatricians should always
rule out organic causes first. Some of these causes like malabsorption,
formula intolerance, genetic disorders, endocrine disorders, are
more common than others like severe OSA with adenotonsillar
hypertrophy. OSA with comorbid adenotonsillar hypertrophy is
known to increase the risk of FTT in infants and children [4].
Case Presentation
Patient information
This is an 8-month-old, former 34 weeks premature, female who
was admitted to the hospital for FTT and severe developmental delay.
Her mother reported that she had constant nasal congestion and
runny nose that never cleared up. Multiple formula changes had been
made with no improvement in her frequent vomiting. Rice cereal was
added to her feeds with no improvement. She had presented to the
ER for bronchiolitis 2 weeks earlier and was treated with albuterol
and antibiotics.
Family history was positive only for mild allergy and constipation.
Patient was on no medication, she did not have any known allergy
and her immunization was up-to-date.
Physical examination
Patient appeared poorly developed with hypotonia and
developmental delay, head lag, and inability to sit up (developmental
age 2-3 months, developmental quotient 38%). Her weight was below
the 1st percentile for her age, and her height was at the 5th percentile
for her age. She was in no acute distress, had nasal congestion, coarse
breath sounds bilaterally, with no wheezing or crackles. She did not
have micrognathia, cleft lip or cleft palate, or any other craniofacial
abnormality. During her first night in the hospital she was found to
have snoring and hypoxia during sleep.
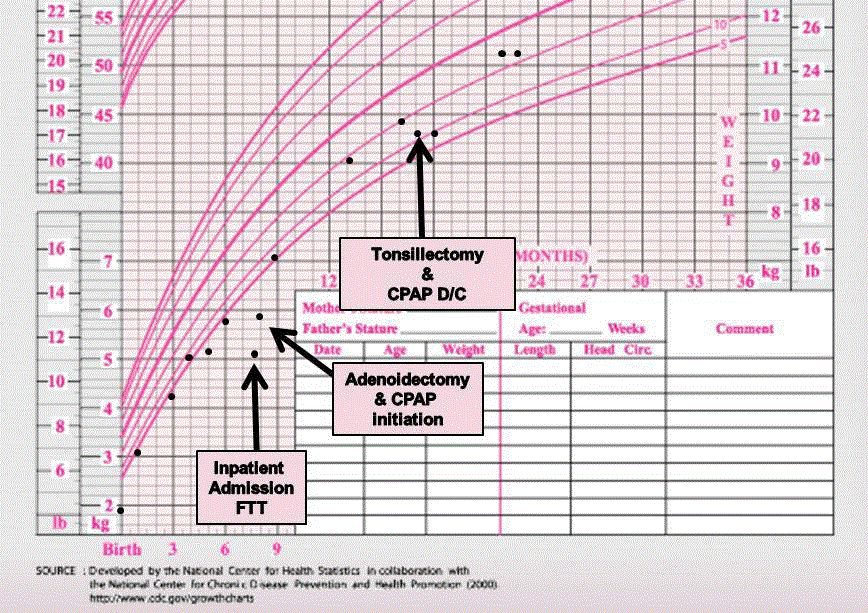
Figure 1
Diagnosis
The patient was started on O2 via nasal cannula (NC) waiting for her surgery. On day 4 of admission, she had a Micro Laryngoscopy, Bronchoscopy, and Adenoidectomy with final diagnosis of 100% obstructing adenoids and mild tracheobronchomalacia. She continued to have snoring and hypoxia during sleep following surgery. Her symptoms were relieved temporarily by changing her body position during sleep. There was oxygen requirement only during sleep. A comprehensive work-up was obtained including sweat chloride test, upper gastrointestinal series, EKG, chest x-ray, brain MRI, and apnea monitor. Sweat chloride test, EKG and brain MRI were normal. Chest x-ray initially showed peribronchial thickening and scattered atelectasis then normalized 10 days later in a repeat film. The upper gastrointestinal series resulted in normal anatomy with multiple episodes of gastroesophageal reflux disease (GERD) to the mid to upper esophagus. She was treated with nasal steroids and ranitidine. The later was changed to omeprazole with no improvement in symptoms. An initial speech evaluation showed no aspiration but deep level penetration with thin liquids and liquids thickened to nectar consistency. The patient was fed with liquids thickened to honey consistency to decrease the risk of aspiration. Multiple apnea monitor downloads were obtained and showed no central apneas but multiple episodes of desaturation mainly in high 70`s to high 80`s. The pattern of the tracing suggested the possible presence of increased work of breathing or upper airway obstruction. Due to the limitation of smart monitor in detecting airway obstruction, clinical correlation and additional testing were recommended to rule out upper airway obstruction. As a result, on day 12 of admission, a Polysomnography (PSG) was performed and it showed severe OSA with an Apnea/Hypopnea index (AHI) of 160.8/ hr (Normal AHI is less than 2/hr for children), an AHI of 236.6/hr in active sleep, and a minimum SpO2 of 65%.
Intervention
On day 16 of admission, a PSG with oxygen titration was done and showed improved oxygenation but worsening of ventilation and much disrupted sleep with AHI of 160/ hour. After discussion with ENT and Pulmonary, on day 20, continuous positive airway pressure (CPAP) treatment was initiated and then titrated progressively as patient started tolerating the interface. She had mildly enlarged tonsils, but surgery was postponed due to concern for postoperative complications in view of age of the patient and the severity of OSA.
Course
Patient showed major clinical improvement with no more
hypoxia at night. Her weight increased by 1 kilogram in 30 days.
She also showed significant developmental gains: she was able to
hold her head up independently, reach for and play with toys, sit up
with minimal assistance, and pull to stand with minimal assistance
(developmental age 6-7 months, developmental quotient 67%), all
of which she was not able to do on admission (Figure 1). She was
discharged on a CPAP of 10cmH2O after 30 days in the hospital.
Patient was followed in sleep clinic regularly after discharge. She
was seen after 1 month, then every 3 months. Patient was doing better
on CPAP and continued to improve her development and weight
gain.
At the age of 14 months, she was standing with support and
babbling (developmental age 11 months, developmental quotient
78%). At the age of 17 months, she had grown from below the 1st
percentile to the 45th percentile for her age, her developmental age
was 13-14 months, and developmental quotient 80%.
One year later, she continued to improve but with symptoms of OSA
when off the CPAP. Tonsillectomy and uvulopalatopharyngoplasty
were performed. After surgery, PSG was repeated. The patient was
2 years old at that time. PSG without intervention showed normal
breathing with an AHI of 0.4/hour. Her swallow study was also normal
and she had no GERD symptoms. CPAP and reflux medications were
stopped and she was allowed to drink liquids of thin consistency. She
was then discharged from sleep clinic due to resolution of her OSA
(Figure 1).
Discussion
Prematurity, GERD, aspiration, tracheobronchomalacia and
laryngomalacia are well known risk factors for OSA and in our
patient could be contributing to the severity of her symptoms [5].
Adenotonsillar hypertrophy is a common cause of OSA in children
between the ages of 2 and 8 years. In our patient, adenotonsillar
hypertrophy lead to OSA at a much younger age and this may be
related to the presence of the other contributing causes. Infants with
OSA are at increased risk of FTT, and developmental and behavioral
compromise.
OSA comorbid adenotonsillar hypertrophy is thought to
contribute to FTT by (1) feeding disorder including feeding fatigue
[6] (2) increased sleep energy expenditure [7,8] and (3) Reduced Growth Hormone [9,10]. In our patient, it is very likely that the
OSA comorbid adenotonsillar hypertrophy also contributed to the
dysphagia and may have caused reduced feeding which in turn may
have contributed to the FTT.
Adenotonsillectomy alone can lead to major improvement in
growth and development in infants with OSA [11]. But infants, like
our patient, that have more than one factor leading to their upper
airway obstruction will not improve with surgery alone if appropriate
work-up and treatment of these factors is not implemented [12,13].
Some patients with OSA do not respond to surgery, and may require more intervention like non-invasive ventilation (NIV)
or occasionally tracheostomy tube placement. Each of these
interventions has their own limitations. NIV with mask carries
an inherent problem of poor adherence and a risk of facial mal
development in children with adequate adherence. Tracheostomy
placement has the potential risk of obstruction and asphyxia in the
absence of immediate intervention, a risk of increased infections and
difficulty with care of the tracheostomy.
NIV is used and studied widely in adults and older children
with OSA. The main problem with NIV is the poor adherence with
treatment. In our patient, we could successfully use NIV for an infant
who was thought to have a greater risk/benefit ratio for surgery
at the time until she could grow and undergo successful surgical
intervention safely.
Our patient also benefitted significantly from being followed
in a sleep clinic on a regular basis where she was re-evaluated for
possibility and safety of surgical intervention that was curative and
led to discontinuation of NIV.
Conclusion
This case report illustrates several important points. First, it underscores the importance of suspecting OSA in infants presenting with FTT and developmental delay. Second, it highlights the possibility of successful use of NIV for treatment of OSA at a very young age. Third, it proves that a close follow-up, a multidisciplinary approach, and repeated evaluation for alternative treatment plans are imperative for long term management of OSA in infants.
Acknowledgements
We would like to acknowledge the sleep team at Arkansas Children’s Hospital for their contribution.
References
- [No authors listed]. Standards and indications for cardiopulmonary sleep studies in children. American Thoracic Society. Am J Respir Crit Care Med. 1996; 153: 866-878.
- Katz ES, Mitchell RB, D'Ambrosio CM. Obstructive sleep apnea in infants. Am J Respir Crit Care Med. 2012; 185: 805-816.
- Chang S, Chae K. Obstructive sleep apnea syndrome in children: epidemiology, pathophysiology, diagnosis and sequelae. Korean J Pediatr. 2010; 53: 863-871.
- Chan J, Edman JC, Koltai PJ. Obstructive sleep apnea in children. Am Fam Physician. 2004; 69: 1147-1154.
- Ramgopal S, Kothare SV, Rana M, Singh K, Khatwa U. Obstructive sleep apnea in infancy: a 7-year experience at a pediatric sleep center. Pediatr Pulmonol. 2014; 49: 554-60.
- Bonuck K, Parikh S, Basilla M. Growth failure and sleep disordered breathing: A review of the literature. International Journal of Pediatric Otorhinolaryngology. 2006; 70950: 769-778.
- Stenlof K, Grunstein R, Hedner J, Sjostrom L. Am J Physiol. 1996; 271 (6 Pt 1), E1036-E1043.
- Li A, Yin J, Chan D, Hui S, Fok T. Sleeping energy expenditure in paediatric patients with obstructive sleep apnoea syndrome. Hong Kong Med. J. 2003; 9: 353-356.
- Selimoglu E, Selimoglu M, Orbak Z. Does adenotonsillectomy improve growth in children with obstructive adenotonsillar hypertrophy?. J Int Med Res. 2003; 31: 84-87.
- Bar A, Tarasiuk A, Segev Y, Philip M, Tal A. The effect of adenotonsillectomy on growth in young children. Otolaryngol. Head Neck Surg. 1991; 104:509-516.
- Freezer NJ, Bucens IK, Robertson CF. Obstructive sleep apnoea presenting as failure to thrive in infancy. J Paediatr Child Health. 1995; 31: 172-175.
- Mitchell RB. Adenotonsillectomy for obstructive sleep apnea in children: outcome evaluated by pre- and postoperative polysomnography. Laryngoscope. 2007; 117: 1844-1854.
- Friedman M, Wilson M, Lin H, Chang H. Updated systematic review of tonsillectomy and adenoidectomy for treatment of pediatric obstructive sleep apnea/hypopnea syndrome. Otolaryngol Head Neck Surg. 2009; 140: 800-808.