Case Report
Primitive Neuroectodermal Tumor of Kidney: A Rare Case Report in an Elderly Lady
Chaitra K1*, Prem Kumar1, Shanta Krishnamurthy2 and Anand2
1Fortis Hospital, Bangalore, India
2SRL Ltd, Bangalore, India
*Corresponding author: Chaitra Krishnagiri, Fortis Hospital, 154/9, Bannerghatta Road, Opposite IIM-B, Bengaluru, Karnataka 560076, India
Published: 07 Sep, 2017
Cite this article as: Chaitra K, Prem Kumar, Shanta
Krishnamurthy, Anand. Primitive
Neuroectodermal Tumor of Kidney: A
Rare Case Report in an Elderly Lady.
Ann Clin Case Rep. 2017; 2: 1433.
Abstract
Primitive Neuroectodermal Tumors (PNET) are malignant, small round cell tumors (RCTs), which commonly de-velop in bone and soft‑tissue of the body. In this article we report a case of Renal PNET that presented in an elderly lady of 85years of age.
Introduction
Primitive Neuroectodermal Tumors (PNET) are malignant, small round cell tumors (RCTs), which commonly develop in bone and soft‑tissue of the body [1,2]. Rarely have they been found in other parts of the body, such as the kidney, bladder, prostate, testis, ovary and uterus, etc [3‑6]. PNET of the kidney are very rare malignant neoplasms. They were first recognized by Stout in 1918 [7]. Because of the similarity of PNET to Ewing’s tumor, it is difficult to estimate their exact number [8]. Until date, about 50 cases have been reported in the literature. These tumors frequently arise in childhood and adolescence with an aggressive clinical course [9]. In this article we report a case of Renal PNET that presented in an elderly lady of 85years of age.
Case Presentation
An 85 year old female patient presented with vague left loin discomfort of a few days duration.
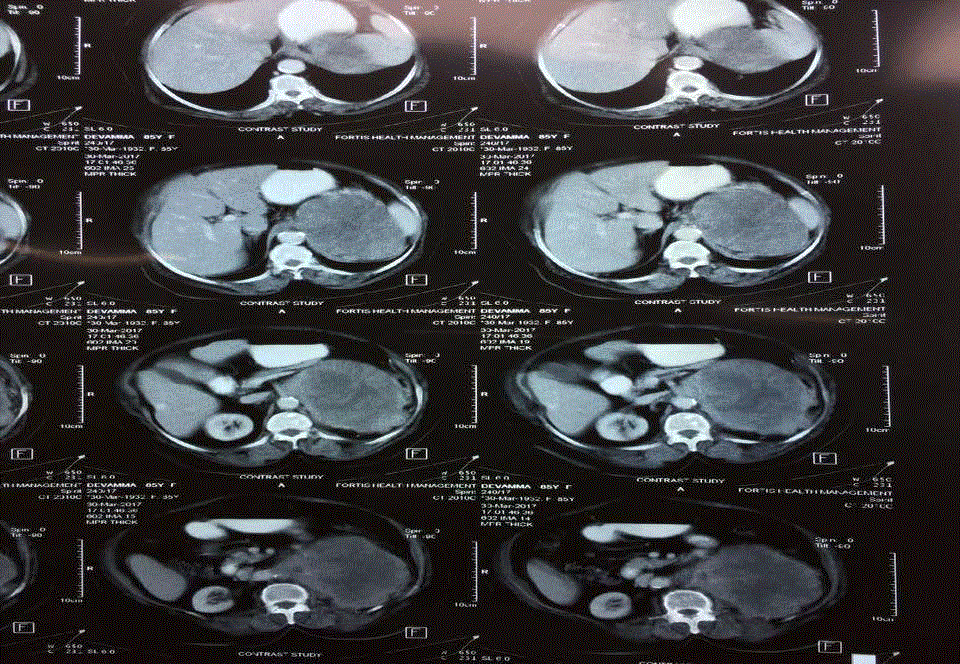
On routine Ultrasonography of Abdomen and pelvis she was found to have a mass lesion in the
left Kidney involving the mid and upper pole, with dimensions- 20 cm × 15 cm × 7 cm. The mass
was reported to be confined to the kidney with no perinephric extension. The diaphragm and the
retroperitoneum were found to be free from any infiltration. A subsequent Contrast enhanced
CT abdomen and pelvis revealed a similar finding with a doubtful lesion in the lower lobe of left
lung. There was no renal vein or IVC involvement or thrombus. Few Lymph nodes were identified
in the renal Hilum. Patient was offered exploratory laparotomy and Left Radical Nephrectomy.
Patient underwent the same on 31/03/2017. Operative findings- A highly vascular mass lesion with
dimensions as per the CT findings, involving the mid and upper pole of left kidney with vascular
and tissue infiltration into the Psoas sheath posteriorly and into the diaphragm was noted. Radical
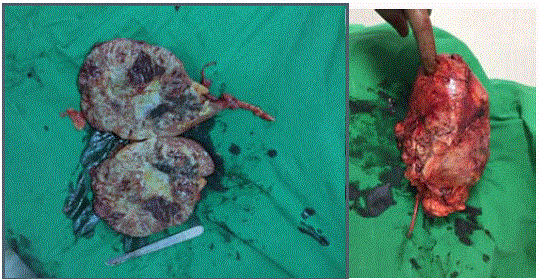
nephrectomy was performed with few hilar lymph nodes. Grossly, the nephrectomy specimen
measured 17 cm × 16 cm × 7.5 cms with large tumor at the upper pole which measured 15 cm ×14cm
× 6.5 cms. The tumor occupied the entire kidney with a very thin rim of parenchyma seen at the
periphery, at the lower pole. The tumor was seen infiltrating the renal sinus, capsule and perirenal
fat. Cut section of the tumor was variegated, grey white with hemorrhagic and necrotic areas. The
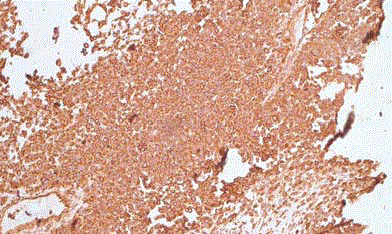
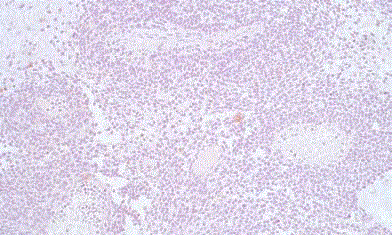
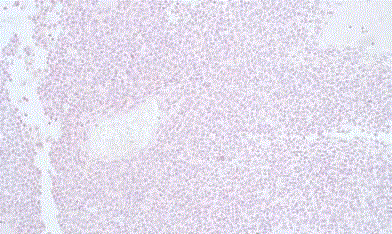
attached ureter appeared unremarkable. Microscopically, it showed an infiltrating malignant tumor
with tumor cells arranged diffusely in sheets with a perivascular preservation of cells and intervening
necrotic areas. Tumor cells demonstrated rounded nuclei with monomorphic appearance. Mitotic
figures were noted with few atypical mitoses. Tumor was seen invading the renal sinus, renal capsule
with extension into the perirenal fat. Ureteric resection margin was free of tumor.
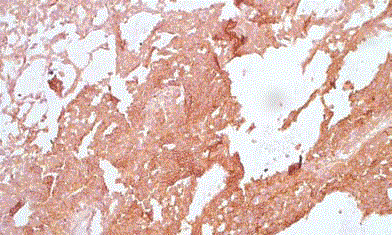
Immunohistochemically (IHC), the tumor cells were positive for Vimentin, CD 99(Mic-2),
NSE and CD 56. They were negative for Wilm’s tumor protein 1, pancytokeratin, synaptophysin
and chromogranin. The Ki-67 index was 10-15%. Based on the morphological features and IHC, a
diagnosis of Renal PNET was made.
Figure 1
Figure 2
Figure 3
Figure 3
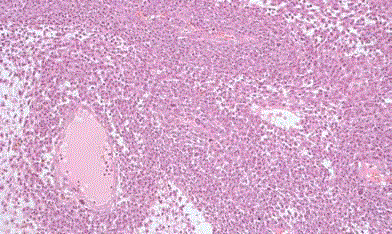
Photomicrograph of H&E stained slide of Renal PNET showing
monomorphic tumor cells with rounded nuclei.
Figure 4
Figure 5
Figure 6
Figure 7
Discussion
Renal PNET appears to be a unique clinical entity that behaves more aggressively than PNET arising at other sites [2]. First described by Stout in 1918, the concept of PNET has evolved to include a group of small round cell malignancies of ubiquitous location and presumed neurectodermal origin. They are defined by the expression of the same protooncogene [10] and the presence of a balanced t (11;22) (q24:q12) chromosomal translocation or a (21:22) rearrangement that results in the fusion of the gene EWS with the FLI1 or ERG gene, respectively [11]. PNET manifests a continuum of neurogenic differentiation, with Ewing’s sarcoma representing the least differentiated and peripheral neuroepithelioma the most differentiated forms [12]. In general, PNET is a very aggressive neoplasm, with 25-50% of patients presenting with metastatic disease, and a 5-year disease free survival rate of 45-55% [13-15]. Renal location of PNET is extremely rare, with only three previously reported cases [16-18] and a small series from the National Wilms Tumor Study (NWTS) published only in abstract form [19]. The largest series of renal PNET is a series from India published by Thyvihally et al, This series included 16 patients who were treated in Tata Memorial hospital. However, the first report came in the year 1994 when Mor et al. [17] described a characteristic renal neoplasia consistent with the diagnosis of malignant peripheral PNET. The male: female ratio is about 3:1. The presenting symptoms and clinical signs are nonspecific and similar to those of other renal tumors. The tumor has been mainly reported in the younger age group with age ranging from 4years to 61 years. However, our patient was much older than this. Of all reported metastatic cases, 44% present with lung metastases only and 51% have a bone or bone marrow involvement with or without lung metastases and 5% present with metastases in other organs.
Conclusion
In conclusion it can be said that renal PNET is an extremely rare peripheral PNET having neural crest origin. They have aggressive clinical course and poor prognosis. It usually presents in adolescents and young adults, but our case was different as it presented in an elderly woman.
References
- Seth A, Mahapatra SK, Saini AK, Biswas B. Primitive Neuroectodermal tumors of kidney: Our experience in a tertiary care center. Indian J Cancer. 2016; 53: 109-112.
- Rodriguez-Galindo C, Marina NM, Fletcher BD, Parham DM, Bodner SM, Meyer WH. Is Primitive Neuroectodermal tumor of kidney a distinct entity?. Cancer. 1997; 79: 2243-2250.
- Seemayer TA, Thelmo WL, Bolande RP, Wiglesworth FW. Peripheral neuroectodermal tumors. Perspect Pediatr Pathol. 1975; 2: 151-72.
- Desai S. Primary primitive neuroectodermal tumour of the urinary bladder. Histopathology. 1998; 32: 477-8.
- Peyromaure M, Vieillefond A, Boucher E, De Pinieux G, Beuzeboc P, Debré B, et al. Primitive neuroectodermal tumor of the prostate. J Urol. 2003; 170: 182-183.
- Rose PG, O'Toole RV, Keyhani-Rofagha S, Qualman S, Boutselis JG. Malignant peripheral primitive neuroectodermal tumor of the uterus. J Surg Oncol. 1987; 35: 165-169.
- Stout AP. A tumor of the ulnar nerve. Proc NY Pathol Soc Proc NY Pathol Soc. 1918; 18: 2-12.
- Casella R, Moch H, Rochlitz C, Meier V, Seifert B, Mihatsch MJ, et al. Metastatic primitive neuroectodermal tumor of the kidney in adults. Eur Urol. 2001; 39: 613-617.
- Gupta NP, Singh BP, Raina V, Gupta SD. Primitive neuroectodermal kidney tumor: 2 case reports and review of the literature. J Urol. 1995; 153: 1890-1892.
- Thiele CJ. Pediatric peripheral neuroectodermal tumors, oncogenes and differentiation. Cancer Invest. 1990; 629-639
- Delattre O, Zucman J, Melot T, Sastre Garau X, Zucker JM, Lenoir GM et al. The Ewing family of tumors.A subgroup of small round cell tumors defined by specific chimeric transcripts. N Engl J Med. 1994; 331; 294-299.
- Dehner LP. Primitive neuroectodermal tumor and Ewing's sarcoma. Am J Surg Pathol. 1993; 17: 1-13.
- Jürgens H, Bier V, Harms D, Beck J, Brandeis W, Etspüler G, et al. Malignant peripheral neuroectodermal tumors. A retrospective analysis of 42 patients. Cancer. 1988; 61: 349-357.
- Marina NM, Etcubanas E, Parham DM, Bowman LC, Green A. Peripheral primitive neuroectodermal tumor (peripheral neuroepithelioma) in children. A review of the St. Jude experience and controversies in diagnosis and management. Cancer. 1989; 64: 1952-1960.
- Kushner BH, Hajdu SI, Gulati SC, Erlandson RA, Exelby PR, Lieberman PH. Extracranial primitive neuroectodermal tumors. The Memorial Sloan-Kettering Cancer Center experience. Cancer. 1991; 67: 1825-1829.
- Chan YF, Llewellyn H. Intrarenal primitive neuroectodermal tumour. Br J Urol. 1994; 73: 326-327.
- Mor Y, Nass D, Raviv G, Neumann Y, Nativ O, Goldwasser B. Malignant peripheral primitive neuroectodermal tumor (PNET) of the kidney. Med Pediatr Oncol. 1994; 23: 437-440.
- Mentzel T, Bultitude MI, Fletcher CDM. Primarer primitive neuroectodermal tumor der niere bei einem erwachsenen. Pathologe. 1994; 15: 124-128.
- Roloson GJ, Beckwith JB. Primary neuroepithelial tumors of kidney in children and adults. A report from the NWTS Pathology Centre (abstract) Mod Pathol. 1993; 6: 67.