Case Report
Genitalia Gangrene Secondary to High Inotropic Support: A Case Report
Jia-Lun Kwok*, Daniel Yong and Yee Mun Lee
Department of Urology, Tan TockSeng Hospital, Singapore
*Corresponding author: Jia-Lun Kwok, Department of Urology, Tan TockSeng Hospital, 11 Jalan Tan TockSeng, 308433, Singapore
Published: 06 Jul, 2017
Cite this article as: Kwok J-L, Yong D, Lee YM. Genitalia
Gangrene Secondary to High Inotropic
Support: A Case Report. Ann Clin Case
Rep. 2017; 2: 1391.
Abstract
We present a rare case report of a gentleman on high dose noradrenaline and vasopressin developing genitalia gangrene.
This is a 54 year old gentleman with pT4N0M1a Recto-sigmoid Cancer and bladder extension post
high Anterior Resection with partial cystectomy three years prior. He developed intra-abdominal
sepsis with shock and was transferred to the intensive care unit with dual high dose inotropes and
developed distal bilateral upper limb gangrene. He was noted on day 6 of inotropic support to have
dry penile-scrotal gangrene with scrotal edema.
Surgical debridement was discussed but family declined in view of the overall poor prognosis and
was thus conservatively managed. The gangrene remained dry and demarcated to the superficial
skin layer with sloughing on day 16of inotropic support.
This suggests a need for vigilance and detailed examination including the genitalia in patients on
high dose inotropes. However, it is still a very rare complication.
Keywords: Infection; Bladder cancer; Inotropes
Abbreviations
ICU: Intensive Care Unit; IV: Intravenous; mcg/kg/min: Micrograms Per Kilogram Per Minute
Introduction
Peripheral limb gangrene is a known complication of high dose inotropic support [1,2] with pathophysiology due to vasoconstriction of distal end vessels resulting in tissue necrosis. This has been reported as a catastrophic complication in the Intensive Care Unit (ICU). However case reports of genitalia gangrene in this setting are very rare. We present a rare case report of a patient on high dose noradrenaline and vasopressin, with complications of upper limb distal hypo-perfusion developing penile-scrotal gangrene.
Case Presentation
Patient background
This is a 54 year old Chinese gentleman with pT4N0M1a Recto-sigmoid Cancer with bladder
extension post high Anterior Resection and partial cystectomy, complicated by a persistent pelvic
collection needing a Hartmann’s operation. He has no other co-morbidities. During the second
year of surveillance, he was found to have colorectal adenocarcincoma recurrence in the bladder
and underwent a Trans-Urethral Resection of Bladder Tumour with subsequent chemotherapy and
radiotherapy.
Clinical history
He presented with fecaluria for 3 days to the outpatient clinic two and a half years after his initial
surgery, and was admitted inpatient. He reported fecal material in the urinary stream associated
with abdominal pain. He was a-febrile and with normal blood pressure. Initial physical examination
revealed the abdomen to be soft and non-tender, renal punch negative bilaterally, and the existing
ileostomy healthy. Grossly feculent urine was noted.
The patient was kept nil by mouth with a naso-gastric tube inserted and placed on low intermittent
suction, with initiation of intravenous (IV) metronidazole and ceftriaxone. An indwelling urinary
catheter was inserted. A computed tomography scan of the abdomen and pelvis was suggestive of an
enterovesical fistula which was also seen during flexible cystoscopy.Subsequently he developedileus.
Bilateral percutaneous nephrostomies were inserted for urinary
diversion; and he was managed with nil by mouth and total parenteral
nutrition with a plan for bowel resection and possible hartmanns
reversal after optimising nutrition.
Unfortunately he developed septic shock secondary to intraabdominal
sepsis on day 17 of admission.The next day, he was
transferred to the ICU and noradrenaline support was started at 0.4
mcg/kg/min.
Blood cultures grew Klebsiella pneumoniae and yeast, with
antibiotics escalated to IV meropenem, amikacin, vancomycin and
fluconazole. His blood pressure improved and noradrenaline was
weaned down to 0.28 mcg/kg/min on ICU day 2. A joint decision
was made with the colorectal service and ICU team for exploratory
laparotomy, adhesiolysis and resection of the entero-vesical fistula.
Post operatively on ICU day 4, he had worsening hypotension
despite being on noradrenaline 1 mcg/kg/min. Vasopressin was started
at 2.4 units/hour with attempts at weaning down noradrenaline. He
underwent a second exploratory laparotomy to rule out ischemic
bowel which returned negative. The patient had multiple episodes of
hypotension and persistent sepsis needing dual high dose inotropes.
On ICU day 5vasopressin was weaned off with noradrenaline at 0.58
mcg/kg/min.
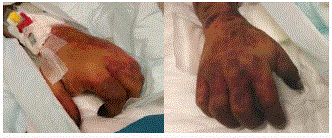
He was noted on day 6 of the ICU admission and inotropic
support during routine physical examination to have new findings
of distal bilateral upper limb duskiness. On further examination
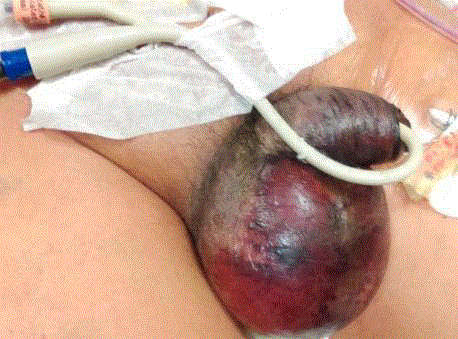
there was genitalia gangrene which was dry, involving the entire
penis and majority of the scrotum with concurrent scrotal edema.
Extent of involvement included the entire penis up to the base, the
entire left hemi-scrotum and a portion of the right hemi-scrotum.
Noradrenaline was at 0.48 mcg/kg/min at that time. This was a
clinical diagnosis, with physical examination showing no evidence
of wet gangrene, pus formation, underlying abscess nor weepy skin
(Figure 1).
Clinical progress
Options for management of the dry scrotal gangrene were
considered, including surgical debridement or conservative
management and wound care with a view of proceeding to surgical
debridement if gangrene progressed to wet gangrene. These options
were discussed, but the family declined surgical debridement in
view of the background of multi-organ dysfunction and the likely
overall poor prognosis. This was thus conservatively managed with
daily dressing change, normal saline cleansing and close monitoring.
Physical examination was done at least twice a day to screen for
progression.
Extent of care was explored in view of the overall poor prognosis
and multi-organ failure. The patient’s wife and family wished for nonescalation
of inotropes and not for cardio-pulmonary resuscitation as
per what the patient wanted.
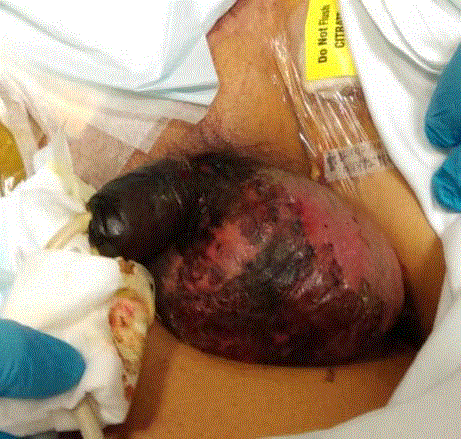
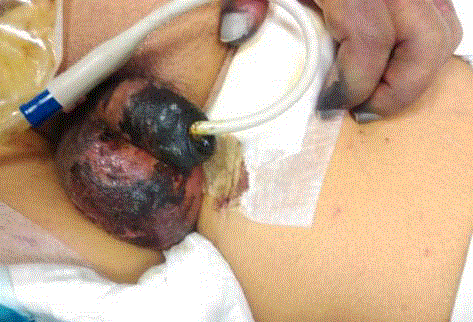
The gangrene remained dry, stable and did not increase in extent.
It started to demarcate to the superficial layer of the scrotum with
sloughing on ICU day 13. Noradrenaline was weaned down to 0.3
mcg/kg/minbut not further (Figure 2). The upper limb distal gangrene
remained dry and demarcated. There was progressive sloughing of
the superficial layer of the scrotum with healthy tissue seen below on
ICU day 16 (Figure 3). Noradrenaline was continued on 0.3 mcg/kg/
min (Figure 4).
Comfort care was maintained with inability to wean down
noradrenaline and the patient became progressively hypotensive
despite fluid challenges. The patient subsequently unfortunately
passed on due to intra-abdominal sepsis on ICU day 20.
Figure 1
Figure 1
Day 6 of inotropic support: Genitalia dry gangrene involving the
entire penis, left hemi-scrotum and portion of the right hemi-scrotum.
Figure 2
Figure 2
Day 13 of inotropic support. Scrotal gangrene demarcation with
sloughing of superficial skin layer.
Figure 3
Figure 3
Day 13 of inotropic support. Bilateral upper limb dry gangrene in
distal digits with no progression.
Figure 4
Figure 4
Day 16 of inotropic support. Scrotal gangrene with sloughing of
the superficial layer and healthy skin underneath. Concurrent left upper limb
digital ischemia which remained dry.
Discussion and Literature Review
A Pubmed search using the key words “scrotum”, “gangrene”,
“inotropes”, “noradrenaline”, “vasopressin” did not reveal any
reported cases of scrotal gangrene associated with inotropic use.
A variety of associations with scrotal gangrene have been reported,
with vasectomy, perianal abscess, circumcisions and inguinal hernia
repairs [1-4] usually reported predominantly in the scrotum and
not the penis. Genitalia gangrene associated with vasculitis has been
reported, but only in the setting of hypersensitivity vasculitis and
buerger’s disease [5,6].
The patient in our report lacked risk factors for peripheral vascular
disease and did not have diabetes mellitus, human immunodeficiency
virus infection, nor was a chronic alcoholic [7]. The likely
pathophysiology in this patient would be obliterative endarteritis of
the subcutaneous arteries contributed by vasoconstriction from high
dose inotropes. Noradrenaline as an alpha-receptor stimulator is used
frequently in patients with septic shock and its vasospastic effect on
end arteries can cause decreased perfusion causing distal ischemia.
This is supported by the gangrene distribution involving several
end arteries with known good blood supply: the scrotum by the
external pudendal and perineal artery, and penis from the superficial
perineal artery and internal pudendal artery [8]. This supports the
pathophysiology of a general vasospastic effect on several end
arteries. However we acknowledge that the background of septic
shock, hypotension and multi-organ dysfunction had also likely
contributed with decreased peripheral perfusion, and compounded
by vasoconstriction from inotropes.
Our patient was on a high dose of noradrenaline up to 1 mcg/
kg/min, with a total duration of 6 days, and the lowest dose at 0.3
mcg/kg/min prior to the development of peripheral gangrene. This
definition is consistent with other intensive care studies where high
dose inotrope was defined as receipt at any point of ≥ 1 mcg/kg/min
of norepinephrine equivalent [9]. The high doses of inotropic support
in our patient was also similar to another case report of four limb
peripheral gangrene where noradrenaline was up to 0.5 mcg/kg/min
[10,11]. Our patient also had concurrent peripheral limb gangrene,
supporting the aetiology of end artery ischemia.
Our diagnosis of dry penile-scrotal gangrene is a clinical one,
based on physical examination. Differentials would include wet
gangrene, underlying abscess or rapidly progressing Fournier’s
gangrene. Our examination revealed that the gangrene was dry, and
noting importantly neither underlying bogginess nor pus within the
underlying subcutaneous layers in the penile shaft and the scrotum,
excluding wet gangrene and underlying abscess. In view of this, further
imaging with ultrasound was not necessary. Close monitoring was
instituted to ensure no further extension of the gangrene boundaries
to exclude a rapidly progressing Fournier’s gangrene.
In terms of management of genitalia gangrene, options included
surgical debridement or conservative management. Deciding factors
to be considered include the gangrene being dry and not wet in nature,
extent of the gangrene and also the overall prognosis of the patient.
The risks of surgical debridement include further hypotension with
general anaesthesia in a patient who is septic, and procedural risks including bleeding and injury to underlying structures. Merits of early
surgical debridement would include early control and preventing
further development into wet gangrene or subsequent secondary
infection which may lead to Fournier’s gangrene. The merits of a
conservative approach would be avoidance of general anaesthesia and
further hypotension, with close observation to allow early decision to
abandon conservative management and opt for surgical debridement
if the gangrene progressed or with evidence of secondary infection.
Our patient had dry gangrene which was muchlocalised to the penis
and scrotum, allowing the option of a conservative approach. The
gangrene also subsequently demarcated and remained dry, with the
superficial layer sloughing off, revealing healthy skin underneath. In
addition, in view of the overall poor prognosis of the patient and in
line with the family and patient’s wishes, the conservative approach
was appropriate with overall goals of care.
Conclusion
Despite genitalia gangrene being rare in a setting of high inotropic use, this case report suggests a need for vigilance and detailed examination including the genitalia in patients on high dose inotropes with septic shock. Surgical debridement may need to be considered early if there are signs of wet gangrene or rapid extension. A multidisciplinary approach involving nursing staff will be crucial in early detection and treatment.
References
- Patel A, Ramsay JW, Whitfield HN. Fournier's gangrene of the scrotum following day case vasectomy. J R Soc Med. 1991; 84: 49-50.
- Lichtenstein D, Stavorovsky M, Irge D. Fournier's gangrene complicating perinal abscess: report of two cases. Dis Colon Rectum. 1978; 21: 377-379.
- Dinc T, Kayilioglu SI, Sozen I, Yildiz BD, Coskun F. Fournier's Gangrene as a Postoperative Complication of Inguinal Hernia Repair. Case Rep Surg. 2014; 2014: 408217.
- Galukande M, Sekavuga DB, Muganzi A, Coutinho A. Fournier's gangrene after adult male circumcision. Int J Emerg Med. 2014; 7: 37.
- Sohn M, Kistler D, Kindler J, Lutzeyer W. Fournier's gangrene in hypersensitivity vasculitis. J Urol. 1989; 142: 823-825.
- Aktoz T, Kaplan M, Yalcin O, Atakan IH, Inci O. Penile and scrotal involvement in Buerger’s disease. Andrologia. 2008; 40: 401-403.
- Bhatnagar AM, Mohite PN, Suthar M. Fournier's gangrene: a review of 110 cases for aetiology, predisposing conditions, microorganisms, and modalities for coverage of necrosed scrotum with bare testes. N Z Med J. 2008; 121: 46-56.
- Dwyer M, Salgado C, Lightner D. Normal Penile, Scrotal, and Perineal Anatomy with Reconstructive Considerations. Sem Plastic Surg. 2011; 25: 179-188.
- Brown S, Lanspa M, Jones JP, Kuttler KG, Li Y, Carlson R, et al. Survival after shock requiring high-dose vasopressor therapy. Chest. 2013; 143: 664.
- Chuan Han Ang, Oon Thien Koo, Tet Sen Howe. Four limb amputations due to peripheral gangrene from inotrope use – Case report and review of the literature. Int J Surg Case Rep. 2015; 14: 63-65.
- Colak T, Erdogan O, Yerebakan O, Arici C, Gurkan A. Symmetrical peripheral gangrene and dopamine. Ulus Travma Acil Cerrahi Derg. 2003; 9: 222-224.