Case Report
Intravascular Papillary Endothelial Hyperplasia: Regarding a Case
Moreillo-Vicente L1*, Gimenez-Bachs JM2, Agusti-Martinez A2 and Salinas-Sanchez AS2
1Department of Urology, Hospital General Almansa, Albacete, Spain
2Department of Urology, Hospital General de Albacete, Albacete, Spain
*Corresponding author: Moreillo-Vicente L, Department of Urology, Hospital General Almansa, Albacete, Spain
Published: 03 Apr, 2017
Cite this article as: Moreillo-Vicente L, Gimenez-Bachs JM,
Agusti-Martinez A, Salinas-Sanchez
AS. Intravascular Papillary Endothelial
Hyperplasia: Regarding a Case. Ann
Clin Case Rep. 2017; 2: 1319.
Abstract
We report a case of a patient who presented with right renal colic pain. Complete workup for colic pain was performed and a heterogeneous lesion was found that seemed to originate from the right adrenal gland. Tumor in the right adrenal gland was suspected and the patient underwent radical nephrectomy. Pathological diagnosis of intravascular papillary reports endothelial hyperplasia, one rare benign lesion. The patient is currently asymptomatic and without findings in follow-up visits.
Keywords: Intravascular papillary endothelial hyperplasia; Renal tumor; Nephrectomy
Introduction
Intravascular papillary endothelial hyperplasia (IPEH) was first described as a malignant lesion by Pierre Masson in 1923 [1], who termed it vegetant intravascular hemangioendothelioma and observed its histological likeness to angiosarcoma. Later, Henschen [2] described a similar intravascular endothelial proliferation, interpreted as a reactive process secondary to vascular inflammation and stasis. We present a case of a patient diagnosed with intravascular papillary endothelial hyperplasia of renal vein.
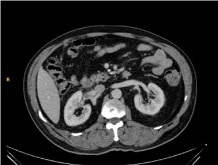
Figure 1
Case Presentation
A 61-year old male patient was referred to our department with symptoms compatible with
right renal colic. The patient had history of hypertension, obstructive sleep apnea syndrome and
benign prostatic hyperplasia treated with alpha-blocker therapy. The only symptom reported by the
patient was intense pain in the right flank, sometimes radiating to the ipsilateral inguinal region.
Physical examination revealed right flank pain with percussion. Results of blood and urine analysis,
abdominal X-ray and ultrasound study were normal. The persistence of symptoms led to the
performance of a CT scan with contrast that revealed a lobulated solid mass sized 3.5 cm × 3 cm × 4
cm, with heterogeneous necrotic core, located at the right renal hilum, and infiltrating the renal vein
(Figure 1). This mass was adjacent to the lower portion of the adrenal gland, to the kidney, and the
posterior portion of the duodenum, and its origin could not be clearly identified. MRI was requested
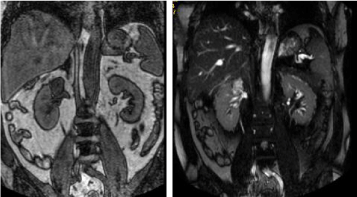
in order to identify the origin, and the mass was described as a tumor both T1-hypointense and T2-
hyperintense (Figure 2), of probable adrenal origin.
Suspecting adrenal injury, hormonal and metabolic workup was carried out with blood and
24-hour urine samples, and the results were: serum cortisol 12.3 μg/dl; 24-h urine cortisol 102.2 ng/dl; DHEA-S: 1.2 μg/ml; 24-h urine dopamine 362 μg/24-h; serum
aldosterone 30 pg/ml; vanillylmandelic acid in urine 5.1; ACTH 11 pg/
ml. MIBG-scintigraphy was also requested but no enhanced lesions
suggestive of pheochromocytoma were found. Surgery was decided
given the findings described and the likelihood of a nonfunctional
adrenal malignancy. Subcostal laparotomy was performed, and
a tumor about 4 cm in diameter was found firmly adhered to the
renal hilum. Simple tumor excision was impossible, so right radical
nephrectomy was carried out. No incidences were reported during
the postoperative follow-up, and the patient was discharged on the
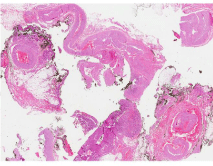
fourth day. During microscopy examination of the surgical specimen,
the kidney and the adrenal gland were unaltered. The mass was close
to the renal hilum and partially contained within a dilated vein, with
thrombosis and endothelial reactive area proliferation (capillary
and papillary), which was compatible with intravascular papillary
endothelial hyperplasia (Figure 3). Visits are scheduled every six
months, and analytical tests and CT images are normal at present.
Figure 2
Figure 2
NMR T1: hypointense image on the right renal hilum. NMR T2:
hyperintense image on the right renal hilum.
Figure 3
Figure 3
Histological specimen. Close to the renal vascular pole and partially
including a dilated vein, an intravascular thrombus and a reactive endothelial
proliferation zone (capillary and papillaroid) are observed.
Discussion
Several morphological features were suggested by Enzinger and
Clearkin [3] that serve in the differential diagnosis of IPEH and
angiosarcoma. These include intraluminal location of the lesion, the
absence of necrotic tissue and the presence of thrombotic material.
These authors also proposed the term intravascular papillary
endothelial hyperplasia. Etiology of IPEH is still unknown, although
trauma has been proposed as the main etiological factor. However,
traumatic history is exceptional. Several authors agree with Clearkin
and Salyer, who believe the cause to be an alteration in the thrombosis
process, consisting of an unusual and peculiar way of thrombus
organization [4,5].
IPEH can occur at any age, and is more frequent in female
patients. The lesions are most often localized in skin vessels, head and
neck, where they appear as small hard masses in the skin of bluish
red color. However, less frequent locations such as jejunum, central
nervous system, liver, and lungs have been reported [5,6].
Three different types of IPEH have been described: Primary
IPEH affecting dilated vascular lakes; secondary or mixed IPEH, after
a preexisting vascular lesion such as hemangioma, arteriovenous
malformations or pyogenic granuloma; and extravascular IPEH,
less frequent and presenting as the result of a hematoma [7]. IPEH
in the renal vein is rare, and very few cases have been reported in
the literature. Its symptoms are variable, ranging from the incidental
finding in an otherwise asymptomatic patient, to colic pain and
hematuria. When imaging techniques are used, CT scan with
contrast reveals a solid lesion with heterogeneous contrast uptake,
and R a lesion hypointense on T1 and hyper intense on T2. These
observations do not exclude malignancy [8,9].
Kidney preservation was possible in one of the cases reported in
the literature [10,11] after preoperative diagnosis and lesion excision.
However, this was not possible in most cases, as malignancy could
not be ruled out, so radical surgery is usual: proximity to the renal
vessels poses technical impossibility, and the suspicion of malignancy
recommends it. Cases with metastases or malignant degeneration
have not been reported.
Preoperative diagnosis of IPEH is challenging because there are
no characteristic symptoms and no imaging technique allows for
adequate differential diagnosis [12]. The latter is very limiting, since
several neoplastic and non-neoplastic lesions can be found in the renal
hilum, including renal carcinoma, angiomyolipoma, schwannoma,
myelolipoma, hemangiopericytoma, lymphoma, cysts, Castleman
disease or lipomas. Therefore, this condition, although rare, must be
considered among the diagnostic possibilities, especially if the origin
is elucidated or proximity to vessels is detected.
References
- Masson P. Hemangioendothelioma vegetant intra-vasculaire. Bull Soc Anat Paris. 1923;93:517-23.
- Henschen F. L’endovasculite proliférante thrombopoïetique dans la lesion vasculaire locale. Ann anat Pathol. 1932;9:113-21.
- Clearkin KP, Enzinger FM. Intravascular papillary endothelial hyperplasia. Arch Pathol Lab Med. 1976;100(8):441-4.
- Johraku A, Miyanaga N, Sekido N, Ikeda H, Michishita N, Saida Y, et al. A case of intravascular papillary endothelial hyperplasia (Masson's tumor) arising from renal sinus. Jpn J Clin Oncol. 1997;27(6):433-6.
- Pelosi G, Sonzogni A, Viale G. Intravascular papillary endothelial hyperplasia of the renal vein. Int J Surg Pathol. 2011;19(4):518-20.
- Salyer WR, Salyer DC. Intravascular angiomatosis: development and distinction from angiosarcoma. Cancer. 1975;36(3):995-1001.
- Hashimoto H, Daimaru Y, Enjoji M. Intravascular papillary endothelial hyperplasia. A clinicopathological study of 91 cases. Am J Dermatopathol. 1983;5(6):539-46.
- Kuo T, Sayers CP, Rosai J. Masson's "vegetant intravascular hemangioendothelioma:" a lesion often mistaken for angiosarcoma: study of seventeen cases located in the skin and soft tissues. Cancer. 1976;38(3):1227-36.
- van den Bogaert S, Boel K, van Poppel H, R. Oyen, B Van Damme. Masson’´s tumour of the kidney. Cancer Imaging. 2002; 2:116-9.
- Logroño R, Sánchez L, Almonte A. Hiperplasia papilar endotelial intravascular: primer caso reportado en la literatura médica dominicana y revision bibliográfica. Acta Med Dom.1993;15:212-216.
- Akhtar M, Aslam M, Al-Mana H, Bamefleh H, Al-Khateeb SS, Lindstedt E. Intravascular papillary endothelial hyperplasia of renal vein: report of 2 cases. Arch Pathol Lab Med. 2005;129(4):516-9.
- Garber BB, Prestipino AJ, Pollack HM, Levine SR, Whitmore KE. Masson's tumor of the kidney: a new renal lesion. J Urol. 1990;143(2):344-6.