Case Report
Unusual Cause of Disseminated Necrotizing Infection of the Abdomenal Wall and Viscera
Hassan Adnan Bukhari1* and Ahmed Kamel Ghanem2
1Department of Surgery, Umm AL-Qura University, Saudi Arabia
2King Abdulaziz Hospital, Saudi Arabia
*Corresponding author: Miss Molly Jakeman, Bolton NHS Foundation Trust, Minerva Road, Farnworth, Bolton, BL4 0JR, United Kingdom
Published: 23 Nov, 2016
Cite this article as: Bukhari HA, Ghanem AK. Unusual
Cause of Disseminated Necrotizing
Infection of the Abdomenal Wall and
Viscera. Ann Clin Case Rep. 2016; 1:
1185.
Abstract
It is uncommon for necrotizing fascitis of the abdominal wall to present in a disseminated manner, extending to most of the intra abdominal viscera. It is also uncommon for the causative organism to be fungus. Usually, it is either mono microbial or polymicropial cause. The usual presentation of this disease entity is changes in skin color from red-purple to patches of blue-gray associated with deeper tissues involvement namely fascia and muscles without involvement of abdominal viscera, associated with picture of toxemia and septic shock. Special staining should be used to identify fungal infestation. This could be the first case reported in the literature among young patients. High index of suspicion and early administration of anti-fungal therapy is needed in unusual presentation of necrotizing infection in young healthy patient.
Introduction
Disseminated necrotizing infection secondary fungal infection is extremely rare. This case
describes a patient with very aggressive necrosis of the abdominal wall the intraabdominal viscera.
The patient presented with septic shock and gangrenous area of the anterior abdominal wall. The
provisional diagnosis was necrotizing fascitis (NF) secondary to bacterial infection, which was
thought to be limited to the abdominal wall, but it turned to involving the whole abdominal wall and
extending to the most of the intraabdominal organs. Histopathology report revealed disseminated
fungal infestation causing tissue necrosis. The patient underwent extensive but damage control
procedure. Despite aggressive supportive care and broad antibiotic therapy, patient expired 12
hours postoperatively.
Necrotizing soft tissue infection is a rare but potentially fatal infection involving skin,
subcutaneous tissue and muscle [1]. Typically, it begins minor trauma, as simple as simple
contusion, minor burn, or insect bite. The disease occurs more frequently in diabetics, alcoholics,
immunosuppressed patients, intravenous drug users, and patients with peripheral vascular
disease [2]. It is commonly associated with systemic inflammatory response syndrome (SIRS) and
septic shock. Patients need aggressive debridement and prolonged intensive care support [3,4].
Necrotizing fasciitis is characterized by widespread necrosis of the subcutaneous tissue and fasciae.
However, it can involve deep fascia and muscle [5]. Although it can occur in any region of the body, abdominal wall, perineum and extremities are the most common sites [6]. The disease progresses rapidly and it is usually caused by polymicrobial symbiosis and synergy [1,4]. Monomicrobial infection usually affects immunocompromised patients (e.g. patients with cancer, diabetes mellitus,
vascular insufficiencies or organ transplantation) [7]. Many aerobic and anaerobic pathogens
may be involved, including but not limited to bacteroides, clostridium, peptostreptococcus,
enterobacteriaciae and pseudomonas. Group A hemolytic streptococcus and staphylococcus aureus,
alone or in synergism, are the initiating infecting bacteria [8]. Fungal infection although reported, is not a common finding in necrotizing fasciitis; only a handful of case reports are available in
literature and mainly in pediatric patients or elderly [9,10].
Treatments include rapid radical debridement and administration of appropriate antibiotics.
However, even with proper treatment, the mortality rate remains as high as 53% [11]. Because NF is a surgical emergency and a life-threatening condition, Aggressive resuscitation, broad antibiotic
administration, intensive care unit (ICU) admission, and immediate surgical debridement are
warranted [12].
Case Presentation
A 35-year-old gentleman referred from another hospital as case
of acute renal failure. The patient has only history of hiccup for
4 days and noticed a small grayish-blue area in umbilical region,
which increased in size with time to reach about 25 cm in maximum
diameter and associated with pain and firmness of the skin. The
patient has unremarkable medical and surgical history, and has no
allergy to any medication.
On arrival to our institution, the patient was in septic shock on
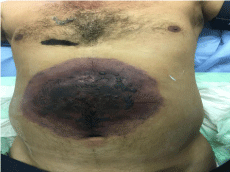
inotropic support but conscious and communicating well. Abdominal
examination reveled a large blackish area about 25 to 30 centimeter
(cm) extending from the epigastric area down to 2cm below the
umbilicus (Figure 1). Abdomen was distended, very tender and rectal
examination revealed altered blood. Laboratory workup showed
leukocytosis and acute renal and hepatic failure. Radiographic
investigation including ultrasound (US) and computerized
tomography (CT) scan of the abdomen revealed fat stranding in the
abdominal wall and mesentery with free fluid.
The patient was immediately transferred to the operating room
with a working diagnosis of necrotizing fasciitis of the abdominal
wall. Extensive abdominal wall necrosis involving skin subcutaneous
fat fascia and muscles was apparent but without pus collection and no
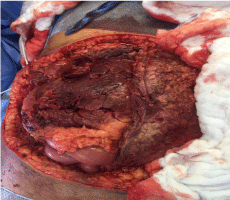
bad odor, which is usually associated with this disease. Upon entering the abdominal cavity, it became clear that there is patchy necrosis of
many organs including omentum, stomach, small and large intestine,
and gallbladder (Figure 2). Limited resection of the dead parts was
carried out. There was evidence of retroperitoneal nercosis too.
Patient was transferred to ICU in unstable condition and planned for
second look next day.
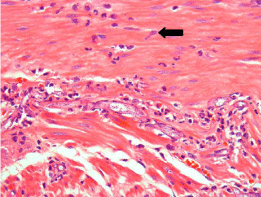
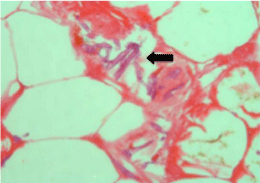
Histopathology report showed non-suppurative necrosis with
fungal invasion, which was variable in size, non-separated hyphae
with 90-degree branching consisting with phycomycosis and vascular
invasion is noticed (Figure 3 and 4).
Figure 1
Figure 2
Figure 2
Intraoperative picture showing excision of the necrotic anterior abdominal wall with gangrenous omentum mesentery and part of the intestine.
Figure 3
Figure 4
Discussion
Subcutaneous phycomycosis was first described in Indonesia in
1956 [13]. It is a saprophytic fungus present in soil, decaying fruit and
vegetable matter as well as in the gut of amphibians and reptiles. It
can cause a variety of clinical manifestations including subcutaneous
zygomycosis, gastrointestinal zygomycosis and occasionally an
acute systemic illness. Subcutaneous zygomycosis is the commonest
presentation reported from many tropical countries including India
[14].
Histopathologically it is a granulomatous infection of the skin
and subcutaneous tissues characterized by the formation of firm and
non-tender swellings, generally on extremities, trunk and rarely other
parts of the body [15]. The disease usually occurs in children, less often
in adolescents and rarely in adults. Males are much more frequently
affected than females [15]. In our study, the patient was a healthy
adult who developed a firm mildly painful bluish nodule over the abdominal wall, which was rapidly progressive over a period of 4 days complicated by intraabdominal involvement and multi-organ failure.
Most patients with phycomycosis respond very well to oral potassium
iodide therapy as well as azoles family particularly itraconazole [14].
This patient was in critical condition postoperatively and he expired
in less than 12 hours from presentation. Starting anti-fungal therapy
early could have changed the outcome. In the absence of classical
features of necrotizing fasciitis in health patient, high index of
suspicion is warranted to start anti-fungal therapy empirically as early
as possible.
References
- Morgan MS. Diagnosis and Management of Necrotizing Fasciitis: A Multi-Parametric Approach. J Hosp Infect. 2010; 75: 249-257.
- Fontes Jr RA, Ogilvie CM, Miclau T. Necrotizing Soft-Tissue Infections. J Am Acad Orthop Surg. 2000; 8: 151-158.
- The Working Group on Severe Streptococcal Infections. Defining the Group A Streptococcal Toxic Shock Syndrome: Rationale and Consensus Definition. JAMA. 1993; 269: 390-391.
- Levine EG, Manders SM. Life-Threatening Necrotizing Fasciitis. Clin in Dermat. 2005; 23: 144-147.
- Tang S, Ho PL, Tang VW, Fung KK. Necrotizing Fasciitis of a Limb. J Bone Joint Surg. 2001; 3: 709-714.
- Green RJ, Dafoe DC, Raffin TA. Necrotizing Fasciitis. Chest. 1996; 110: 219-229.
- Gugnani HC. A Review of Zygomycosis due to Basidiobolus Ranarum. Eur J Epidemiol. 1999; 15: 923-929.
- Sarani B, Strong M, Pascual J, Schwab CW. Necrotizing Fasciitis: Current Concept and Review of the Literature. J Am Coll Surg. 2009; 208: 279-288.
- Newton WD, Cramer FS, Norwood SH. Necrotizing Fasciitis from Invasive Phycomycetes. Crit Care Med. 1987; 15: 331-332.
- Lakshmi V, Rani TS, Sharma S, et al. Zygomycotic Necrotizing Fasciitis Caused by Apophysomyces Elegans. J Clin Microbiol. 1993; 31: 1368-1369.
- Jarrett P, Rademaker M, Duffill M. The Clinical Spectrum of Necrotising Fasciitis: A Review of 15 Cases. Aust N Z J Med. 1997; 27: 29-34.
- Maynor M: Necrotizing Fasciitis 2009. 1-20.
- Burkitt DP, Wilson AM, Jelliffe DB. Subcutaneous Phycomycosis: A Review of 31 Cases Seen in Uganda. Brit Med J. 1964; 1: 1669-1672.
- Sujatha S, Sheeladevi C, Khyriem AB, Parija SC, Thappa DM. Subcutaneous Zygomycosis Caused by Basidiobolus Ranarum: A Case Report. Indian J Med Microbiol. 2003; 21: 205-206.
- Gugnani HC. A Review of Zygomycosis due to Basidiobolus Ranarum. Eur J Epidemiol. 1999; 15: 923-929.