Case Report
Myocardial Infarction after Transurethral Resection of the Prostate Surgery
Peng Zhendan and Yin Ning*
Department of Anesthesiology, Zhongda Hospital, Southeast University, China
*Corresponding author: Yin Ning, Department of Anesthesiology, Zhongda Hospital, Southeast University, Nanjing, China
Published: 19 Sep, 2016
Cite this article as: Zhendan P, Ning Y. Myocardial
Infarction after Transurethral Resection
of the Prostate Surgery. Ann Clin Case
Rep. 2016; 1: 1142.
Abstract
We report the case of a 67-year-old man with hypertension and coronary heart disease who underwent transurethral resection of the prostate surgery with chest tightness, continuous palpitation, dyspnea, 49 hours after back to ward. An electrocardiogram confirmed an inferior and an anterolateral ST depression myocardial infarction. Cardiac troponin I, (cTnI) was 0.08 ng/ml. This is typically acute myocardial infarction after non-cardiac surgery. So he was shifted to cardiologist department for further treatment and discharged two weeks later when he felt fine but was presented to emergency room and expired for acute heart failure one month after discharge.
Keywords: Myocardial infarction; Transurethral resection of the prostate surgery
Introduction
Patients undergoing non-cardiac surgery are at risk of postoperative major vascular complications (i.e., vascular death, non-fatal myocardial infarction (MI), non-fatal cardiac arrest, and non-fatal stroke), and MI is the most common major complication [1]. Antiplatelet therapy should be continued as long as one lives for secondary prevention of acute myocardial infarction. But when it comes to a surgical scenario the dilemma goes to every doctor for concerning about the bleeding if the antiplatelet therapy continued or thrombogenesis when the antiplatelet therapy was discontinued. Although there’s lots of guidelines which strongly recommended the continue of antiplatelet therapy [2-4] and restarted as soon as possible [5]. There are still concerns of bleeding for surgeons especially for prostate surgery beyond second vascular complications perioperatively [6]. Transurethral prostatectomy (TURP) is a common urological procedure which can be associated with considerable blood loss. For this reason the use of antiplatelet and anticoagulant therapy poses a management dilemma for urologists. Significant blood loss alters the risk-benefit ratio and many urologists and patients consider that the increased morbidity and possible mortality negate the potential benefits [7]. We illustrate a case that the patient end up dead after the second attack of AMI after TURP which the antiplatelet therapy was discontinued before the surgery and was not restarted as soon as possible.
Case Presentation
A 67 year old man with progressive difficulty of micturition for ten years and now presented
with dysurine and urine pain for a week was admitted for selective transurethral resection of prostate
surgery on June 23, 2014. He has a remote history of hypertension but under control with medicine,
and underwent percutaneous coronary intervention (a stent in RA) one year ago for chest pain.
Seven days after initial stabilization which included red blood cell transfusion, stopped aspirin
taking for seven days without any bridging treatment and a consultation of cardiologist and
anesthesiologist. The procedure from intubation to extubation was uneventful.
However, 49 hours after the end of the surgery, he was complained of chest pain,
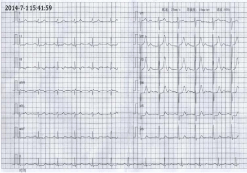
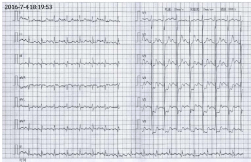
continuous palpitation and could not lie down. The ECG showed an abnormal Q wave in lead II
III AVF and a depression of ST segment in lead V2-V6 (Figure 1 and 2), then nitroglycerin was
pumping but no release of any symptom and cardiac troponin I (TNI) testing was going on which
was turned to be 0.08 ng/ml.
The diagnosis went to ACS. The patient was shifted to Cardiologist department for further treatment. To prevent bleeding, there’s no anti-platelet and anticoagulating
therapy until seven days after surgery. The patient
feels better and coronary angiography was checked and proved a
new stenosis in right coronary artery so a stent was placed. He was
discharged when the TNI goes down to 0.03 ng/ml. unfortunately,
one month after discharged, he was admitted again for acute heart
failure and he died within 48 hours.
Discussion
For this case, acute myocardial infarction happened after TURP
surgery which was defined by James Khan etc, in 2014, myocardial
infarction/injury after non-cardiac surgery (MINs) [8].
Perioperative myocardial injury is the most common
cardiovascular complication following non-cardiac surgery [9,10].
Most of the MINs occurred within 48 hours of surgery (74.1%) [11]
of greater importance, the 30-day mortality rate was 11.6% among
those who had an MI versus 2.2% for those who did not (P<.001)
[12]. In contrast to the diagnosis of spontaneous acute myocardial
infarction, the diagnosis of MINs takes into account the silent (90%)
presentation of painless myocardial infarction and is based on cardiac
troponin only, i.e. it does not require the presence of symptoms [8,13].
Diagnostic criteria: Troponin T level ≥ 0.03 ng/ml [13]. MINs in the
POISE Trial demonstrated that two drugs (i.e., ASP and a statin) were
associated with a statistically significant, risk-adjusted reduction in
30-day mortality [14]. More treatment at 9-12 days after surgery is
median days for decreasing the 30-day mortality [15]. To prevent
MINs after non-cardiac surgery, it seems undeniable to continue the
anti-platelet therapy perioperatively.
However, if anti-platelet or anti-coagulant drugs are not stopped
for TURP, there is an unacceptable burden of bleeding. If the drugs
are stopped there is an unacceptable rate of cardiovascular events.
So this issue still needs to be resolved. 2016 ACC/AHA guideline
focused update on duration of dual antiplatelet therapy in patients
with coronary artery disease, there’s class I recommendations that in
patients treated with DAPT after coronary stent implantation who
must undergo surgical procedure that mandate the discontinuation of
P2Y12 inhibitor therapy, it is recommended that aspirin be continued
if possible and the P2Y12 platelet receptor inhibitor be restarted as
soon as possible after surgery [16].
For those patients with cardiac risk factors on low dose aspirin
alone this can be continued in the perioperative period without
increased risk of major bleeding [2,17]. A multicentre, retrospective, observational study demonstrated that perioperative antiplatelet
discontinuation was the strongest independent predictor of 30-
day major adverse cardiac events (MACE) (odds ratio [OR] =25.8,
confidence interval [CI] =3.37-198, p=0.002). Perioperative aspirin
(adjusted OR 0.27, 95 % CI 0.11-0.71, p=0.008) was significantly
associated with a lower risk of MACE [3]. Aspirin should be continued
perioperatively in the majority of surgical operations, whereas dual
antiplatelet therapy should not be withdrawn for surgery in the case
of low bleeding risk. In selected patients at high risk for bleeding
and ischaemic events, when oral antiplatelet therapy withdrawal is
required, perioperative treatment with short-acting intravenous
glycoprotein IIb/IIIa inhibitors (tirofiban or eptifibatide) should be
taken into consideration [4].
Conclusion
For patients who need anti-platelet therapy for prevention of the secondary acute myocardial infarction perioperatively, anti-platelet therapy should be continued until the surgery day or treatment with short-acting intravenous anti-coagulant drugs.
Figure 1
Figure 2
References
- Group PS. Effects of extended-release metoprolol succinate in patients undergoing non-cardiac surgery (POISE trial): a randomised controlled trial. Lancet. 2008; 37: 1839-1847.
- Culkin DJ, Exaire EJ, Green D, Soloway MS, Gross AJ, Desai MR, et al. Anticoagulation and antiplatelet therapy in urological practice: ICUD/ AUA review paper. J Urol. 2014; 192: 1026-1034.
- Rossini R, Musumeci G, Capodanno D, Lettieri C, Limbruno U, Tarantini G, et al. Perioperative management of oral antiplatelet therapy and clinical outcomes in coronary stent patients undergoing surgery. Results of a multicentre registry. Thromb Haemost. 2015; 113: 272-282.
- Rossini R, Musumeci G, Visconti LO, Bramucci E, Castiglioni B, De Servi S, et al. Perioperative management of antiplatelet therapy in patients with coronary stents undergoing cardiac and non-cardiac surgery: a consensus document from Italian cardiological, surgical and anaesthesiological societies. Euro Intervention. 2014; 10: 38-46.
- Levine GN, Bates ER, Bittl JA, Brindis RG, Fihn SD, Fleisher LA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2016; 68: 1082-1115.
- Devereaux PJ, Mrkobrada M, Sessler DI, Leslie K, Alonso-Coello P, Kurz A, et al. Aspirin in patients undergoing noncardiac surgery. N Engl J Med. 2014; 370: 1494-1503.
- Taylor K, Filgate R, Guo DY, Macneil F. A retrospective study to assess the morbidity associated with transurethral prostatectomy in patients on antiplatelet or anticoagulant drugs. BJU Int. 2011; 108: 45-50.
- Khan J, P Alonso-Coello, PJ Devereaux. Myocardial injury after noncardiac surgery. Curr Opin Cardiol. 2014; 29: 307-311.
- Mangano DT. Perioperative cardiac morbidity. Anesthesiology. 1990; 72: 153-184.
- Mangano DT, Adverse outcomes after surgery in the year 2001--a continuing odyssey. Anesthesiology. 1998; 88: 561-564.
- Thygesen K, Joseph S Alpert, Allan S Jaffe, Maarten L Simoons, Bernard R Chaitman, Harvey D White. Third universal definition of myocardial infarction. J Am Coll Cardiol. 2012; 60: 1581-1598.
- Landesberg G, W Scott Beattie, Morris Mosseri, Allan S Jaffe, Joseph S Alpert. Perioperative myocardial infarction. Circulation. 2009; 119: 2936- 44.
- Botto F, Alonso-Coello P, Chan MT, Villar JC, Xavier D, Srinathan S, et al. Myocardial injury after noncardiac surgery: a large, international, prospective cohort study establishing diagnostic criteria, characteristics, predictors, and 30-day outcomes. Anesthesiology. 2014; 120: 564-578.
- Devereaux PJ, Xavier D, Pogue J, Guyatt G, Sigamani A, Garutti I, Leslie K, et al. Characteristics and short-term prognosis of perioperative myocardial infarction in patients undergoing noncardiac surgery: a cohort study. Ann Intern Med. 2011; 154: 523-528.
- Biccard BM. Detection and management of perioperative myocardial ischemia. Curr Opin Anaesthesiol. 2014; 27: 336-343.
- Levine GN, Bates ER, Bittl JA, Brindis RG, Fihn SD, Fleisher LA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in Patients with coronary artery disease: a report of the american college of cardiology/american heart association task force on clinical practice guidelines: an update of the 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention, 2011 ACCF/AHA guideline for coronary artery bypass graft surgery, 2012 ACC/AHA/ACP/AATS/ PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease, 2013 ACCF/AHA Guideline for the management of st-elevation myocardial infarction, 2014 AHA/ACC guideline for the management of patients with non-st-elevation acute coronary syndromes, and 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery. Circulation. 2016; 134: e123-155.
- Strosberg DS, orbey T, Henry JC, Starr JE. Preoperative antiplatelet use does not increase incidence of bleeding after major operations. Surgery. 2016; 160: 968-976.