Case Report
Interstitial Pneumonitis after Cadmium Exposure: Is it Reversible?
Virendra Singh1*, Nishtha Singh2, Sheetu Singh3 and Bharat Bhushan Sharma4
11Department of Pulmonary Medicine, Asthma Bhawan, India
2Department of Pulmonary Medicine, Asthma Bhawan, India
3Department of Chest and Tuberculosis, SMS Medical College, India
4SMS Medical College and Hospital, India
*Corresponding author: Virendra Singh, Department of Pulmonary Medicine, Asthma Bhawan, Jaipur, Rajasthan, India
Published: 02 Sep, 2016
Cite this article as: Singh V, Singh N, Singh S, Sharma BB.
Interstitial Pneumonitis after Cadmium
Exposure: Is it Reversible?. Ann Clin
Case Rep. 2016; 1: 1119.
Abstract
Introduction: Cadmium fume generation, during silver-cadmium alloy formation, is a common
occurrence in the silver industry. It can involve various organ systems but its toxic effects have not
been widely reported.
Case Presentation: A silver merchant developed respiratory distress and substernal pain after a
single exposure to cadmium fumes generated during silver-cadmium alloy formation. Chest
X-ray revealed bilateral ground glass haze and computed tomography (CT) also revealed changes
suggestive of interstitial pneumonia. Bronchoalveolar lavage was suggestive of increase in the
number of polymorphonuclear neutrophils. Forced vital capacity (FVC) showed a restrictive
pattern. Oral prednisolone was initiated on the third day of the illness. FVC repeated at the fourth
week showed improvement with return of FVC towards normal. The chest CT scan repeated after
six weeks showed complete resolution with no residual fibrosis.
Conclusion: Even a single brief episode of cadmium fume exposure can cause rapid onset of
pneumonitis. Steroids when started early in course of illness were temporally associated with a good
clinical outcome. Definite studies are however required to establish the role of steroids in cadmium
fume induced interstitial pneumonitis.
Abbreviations
ECG: Electrocardiography; HRCT: High Resolution Computed Tomography; 2D-ECHO: Two Dimensional Echocardiography; SGOT: Serum Glutamic Oxaloacetic Transaminase; SGPT: Serum Glutamic Pyruvic Transaminase
Introduction
Cadmium is a white colored metal with usage in many industries. It is used in making an alloy with silver to improve the malleability of silver. It is preferred over other metals because it can be mixed with silver without changing its color. India is one of the leading consumers of silver and millions of people who work in the silver industry are at risk of developing cadmium toxicity. During the process of alloy formation, cadmium fumes are generated which when inhaled can cause acute and chronic toxic effects on various organ systems including nose, throat, lungs, kidneys, liver, hematopoietic and nervous system and can be carcinogenic [1,2]. We report a patient with cadmium toxicity that developed rapid onset of pulmonary damage in the form of interstitial pneumonitis after a single exposure to cadmium fumes in a closed space. Appropriate patient consent was taken and approval from ethics committee was also obtained.
Case Presentation
A 35 years aged male, non smoker, silver merchant by occupation, resident of Jaipur, Rajasthan
was admitted in the hospital with complaints of respiratory distress. Two days earlier he had been
involved in preparation of silver-cadmium alloy. During the process he was exposed to cadmium
fumes. He had never been exposed to cadmium fumes to the best of his knowledge. The procedure
involved heating of silver and cadmium to a temperature of 1200˙C in an open furnace with
subsequent generation of yellow cadmium fumes. Only two metals, silver and cadmium were used
during the procedure in a ratio of 4:1 respectively. The procedure was carried out in a closed room without any window or exhaust and without any use of any personal
protective by the subject. He completed the procedure in half an hour.
Two and half hours later, the patient developed chest discomfort in
the form of sub-sternal pain and difficulty in breathing. His distress
progressively increased to the extent that the patient was unable to
walk even a few steps. There was no history of fever, chills, headache,
myalgia, metallic taste in mouth and hemoptysis. The patient did not
give previous history of hospitalization or any major illness in the
past. There was no history of exposure to pigeon dust or contaminated
organic material. He took analgesics and bronchodilators by the local
doctor but got no relief. The patient was admitted to the hospital on
the third day. Clinical examination revealed that the respiratory rate
was 32 breaths per minute, blood pressure was 130/80 mm of Hg,
pulse rate was 102 beats per min and fine inspiratory crepitations were
heard on auscultation in bilateral basal lung fields. Routine blood
investigations were normal except that the total leukocyte count was
15730 cells/mm3 (normal range - 4000-11000 cells/mm3), serum urea
was 63 mg/dl (Normal range – 7-21 mg/dl), serum creatinine was 1.0
mg/dl (normal range – 0.5-1 mg/dl), SGOT was 240 U/l and SGPT
was 400 U/l (normal range – 0-42 U/l). Arterial blood gas analysis
on admission (at room air) was as follows: pH = 7.442, PaO2 = 85
mm of Hg, PaCO2 = 46.9 mm of Hg, HCO3- = 32.3 mEq and SpO2
= 96.7%. Blood culture, urine culture and sputum examination were
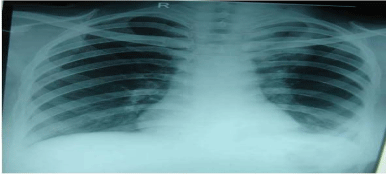
normal. Chest radiograph showed ground glass haze in bilateral
lower lung zones (Figure 1). Electrocardiography (ECG) and two
dimensional echocardiography (2D ECHO) were normal. High
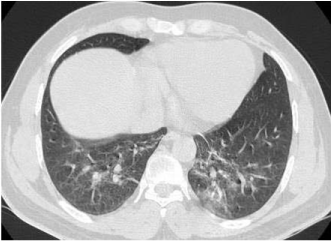
resolution computerized tomography (HRCT) scan of thorax was
done on the day of admission and it revealed ground-glass haziness
with subpleural plate like atelectasis in the peripheral aspects of both
lower lobes, lingular segments of left upper lobe and right middle
lobe (Figure 2). The tracheobronchial tree was normal on inspection
during flexible bronchoscopy. Bronchoalveolar lavage (BAL) was
composed of 63% neutrophils and 24% lymphocytes. There were no
prismatic crystals in the BAL fluid. Blood cadmium levels assessed by
atomic absorption spectrometry on the 4thday of illness were 0.99 μg/ liter (normal levels of cadmium are less than 0.5 μg/liter in blood).
After careful history elicitation and chest CT evaluation a
diagnosis of cadmium induced pneumonitis was made. The history of
exposure to cadmium fumes, onset of symptoms two and a half hours
after exposure, chest CT findings suggestive of interstitial pneumonitis
and raised cadmium levels in the blood were clues to the diagnosis.
He was given oral antibiotics (C. amoxicillin + clavulanic acid 625
mg thrice a day) and oral prednisolone. Prednisolone was given in
the dose of 20 mg once daily in the morning for one month. With
initiation of oral steroids, there was reduction in respiratory rate from
32/min on the 1st day to 25/min on the 2nd day and finally stabilized
to 15-18 /minutes from the 4th day onwards. Clinical symptoms
improved gradually and patient started normal daily activities by
the 14th day. Patient was unable to perform spirometry initially,
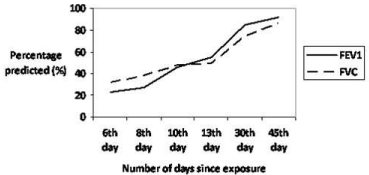
however, repeat testing done on the 6th day of illness demonstrated
restrictive ventilatory defect with a forced vital capacity (FVC) of 1.5L
(32% of the predicted for age and height). Subsequently he showed
improvement in FVC and on day 45 it had increased to 3.9L (83%
of the predicted for age and height as shown in Table 1 & Figure 3).
Blood leukocyte counts, blood urea and liver enzymes returned
to normal levels on the seventh day. Antibiotics were stopped after
microbiological yield was negative. The oral steroids were tapered
after the initial one month with doses of 10mg once daily for five
days, 5 mg once daily for the next five days and 5mg on alternate days
for next five days before completely stopping them (total duration of
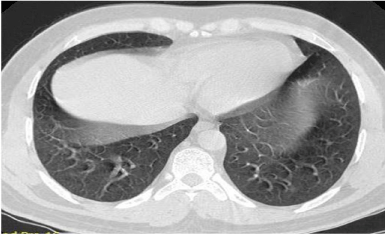
steroid administration– 6 weeks). Repeat HRCT done after one and a
half month of the episode of cadmium exposure showed resolution of
all acute inflammatory reaction, with no evidence of residual fibrosis
or ground-glass opacity (Figure 4).
Figure 1
Figure 2
Results and Discussion
Cadmium toxicity has been previously mentioned in literature
but this case is unique because of the rapidity of onset of interstitial
pneumonitis after a single brief exposure to cadmium fumes and the resolution of clinical symptoms and radiological shadows after a short course of steroids.
Acute exposure to moderate concentrations [200-500 μg/m3] of
cadmium fumes can cause the symptoms of fume fever characterized
by metallic taste, fever, malaise, joint pains, cough, sore throat, chest
tightness and fatigue. The fume fever develops 4-8 hours after exposure
[3,4] and lasts for two to three days. However, very intense exposure
can cause pulmonary edema and pneumonitis [5]. Deaths due to
respiratory failure have also been reported [6]. Persistent impairment
in respiratory functions has been reported following acute and brief
exposure to cadmium fumes [7]. Pulmonary fibrosis in a survivor of
single exposure to cadmium fumes has also been reported [8]. The
onset of illness in our patient was within two and a half hours of
exposure (more rapid than metal fume fever). He worked only for
thirty minutes and did not report any fever, myalgias, metallic taste
in mouth and joint pains which are classical symptoms of metal fume
fever. The patient did not use any personal protective equipment and
the windows of the room were also closed, therefore exposure proved
more intense.
Acute hypersensitivity pneumonitis is another differential
diagnosis in which the patient presents with sudden onset of dyspnea,
fever, myalgias, arthralgias and cough, 2-9 hours after exposure to
contaminated organic matter [9]. In the above mentioned case there
was no history of exposure to any organic material. Also there was
strong temporal association of the symptoms with cadmium fume
exposure.
Cadmium exposure has been known to cause liver damage [10]
which explains the raised enzymes in our patient. But, the primary
organ of damage was however, the lung.
Small case series have been reported regarding the role of steroids
in preventing permanent damage with variable results. Seidal et al.
[6] had reported a patient with cadmium induced pulmonary toxicity
who died despite receiving steroids. However, the steroids were
initiated very late in the course of illness in their case (after ten days of
exposure). Our patient developed similar symptoms after cadmium
fume exposure but the steroids were initiated sooner (third day).
Improvement in symptoms of the patient could be attributed
to the normal return of lung functions after cessation of exposure
to cadmium fumes. Chan et al. [11] had previously studied that
impairment in respiratory functions due to cadmium dust exposure,
was partly reversible if exposure was terminated. However they had
studied a healthy cohort of factory workers exposed to cadmium dust,
who had reduced respiratory functions and no clinical symptoms of
respiratory insult. Our case was unique because he had symptoms
of respiratory distress and radiological evidence of interstitial
pneumonitis. The resolution of symptoms and improvement in lung
functions was much faster with steroids than the resolution in the
cohort of Chan et al. [11]. Thus, steroids are temporally associated
with faster resolution of symptoms of interstitial pneumonitis.
The case report highlights the risk of cadmium toxicity even after
a single exposure and good outcome with early start of steroids. The
role of steroids in treatment of cases of acute cadmium fume exposure
still needs to be researched.
Figure 3
Figure 3
Spirometry values (FEV1 and FVC) as percentage of predicted
values on subsequent days after exposure.
Figure 4
References
- Waalkes MP. Cadmium carcinogenesis. Mutat Res. 2003; 533: 107-120.
- IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Man. Geneva: World Health Organization. International Agency for Research on Cancer, 1972.
- Dickson RP, Schwartz DA. Acute and chronic responses to toxic inhalations. In: Fishman AP, Elias JA, Fishman JA, Grippi MA, Senior RM, Pack AI, editors. Fishman’s pulmonary disease and disorders. United States: Mc Graw Hill. 2008; 1005.
- US department of health and human services. Draft Toxicological profile of Cadmium. Georgia: ATSDR; 2008.
- Beton DC, Andrews GS, Davies HJ, Howells L, Smith GF. Acute Cadmium fume poisoning: Five cases with one death from renal necrosis. Br J Ind Med. 1966; 23: 292-301.
- Seidal K, Jorgensen N, Elinder CG, Sjogren B, Vahter M. Fatal cadmiuminduced pneumonias. Scand J Work Environ Health. 1993; 19: 429-431.
- Barnhart S, Rosenstock L. Cadmium chemical pneumonitis. Chest. 1984; 86: 789-791.
- Townshend RH. Acute cadmium pneumonias: a 17 year follow up. Br J Ind Med. 1982; 39: 411-412.
- Enelow RI. Hypersensitivity pneumonitis. In: Fishman AP, Elias JA, Fishman JA, Grippi MA, Senior RM, Pack AI, editors. Fishman’s pulmonary disease and disorders. United States: Mc Graw Hill, 2008; p. 1162.
- Dudley RE, Svoboda DJ, Klaassen CD. Acute exposure to cadmium causes severe liver injury in rats. Toxicology and applied pharmacology. 1982; 65: 302-313.
- Chan OY, Poh SC, Lee HS, Tan KT, Kwok SF. Respiratory function in cadmium battery workers: A follow-up study. Ann Acad Med Singapore. 1988; 17: 283-287.