Case Report
Reversible Cytotoxic Cortex Oedema Associated with Levofloxacin-Induced Seizures. A Case Report
Roberto Padrini1*, Marco Panfili M2, Marta Martin2, Mariagiulia Anglani3
1Department of Cardiological, Department of Medicine, University of Padova, Italy
2Department of Cardiological, Thoracic and Vascular Sciences, University of Padova, Italy
3Department of Cardiological, Department of Neurosciences, University of Padova, Italy
*Corresponding author: Roberto Padrini, Department of Medicine, University of Padova, Via Giustiniani 2, 35128 Padova, Italy
Published: 23 Aug 2016
Cite this article as: Padrini R, Marco Panfili M, Martin M,
Anglani M. Reversible Cytotoxic Cortex
Oedema Associated with Levofloxacin-
Induced Seizures. A Case Report. Ann
Clin Case Rep. 2016; 1: 1091.
Abstract
Aim: To describe a case of levofloxacin-induced seizures associated with reversible magnetic resonance (MR) brain alterations.
Methods: A 95-year-old white woman had three convulsive episodes two days after starting
levofloxacin therapy (500mg qd). After drug withdrawal, seizures subsided and a diagnostic path
was instituted.
Results: Acute cerebrovascular events were excluded. MR images obtained 7 days after the onset
of seizures showed bilateral alterations in the medial prefrontal cortex, compatible with cytotoxic
oedema. Thirty-seven days later, such alterations had disappeared.
Conclusions: Cytotoxic oedema may have been produced by massive glutamate release during
epileptic episodes. However, a glutamatergic effect of fluoroquinolones cannot be excluded and
deserves further study.
Case Report
A 95-year-old white woman was admitted for severe asthenia and shortness of breath. She
had a clinical history of hypertension and chronic constipation. Blood laboratory data revealed
severe anaemia associated with iron deficiency, which was treated with blood transfusion and
administration of iron i.v. Since potassium serum levels were below the lower normal limit (2.8
mmol/L), oral therapy with a K-sparing diuretic was instituted (canrenone 50mg qd). Haematochemical
indexes of liver and renal function were within normal ranges; in particular, eGFR was
82 mL/min/1.73m2 (CKD-EPI formula). After suspected occult intestinal bleeding, endoscopic
examinations revealed colon diverticulosis, particularly severe in the distal tract. After the above
treatments, haemoglobin rose to 11.5mg/dL, potassium to 3.5 mmol/L, and respiratory symptoms
disappeared.
On the last day of hospitalisation, the patient developed cystitis, so that oral treatment with
levofloxacin 500mg qd was started in hospital and continued at home. Other drugs prescribed at
discharge were: esomeprazole 20mg qd (8 a.m.), rifaximin 400mg bd (8 a.m. /4 p.m. for 7 days),
canrenone 50 mg qd (2 p.m.), furosemide 25mg qd (8 a.m.), allopurinol 300mg qd (8 a.m.),
amlodipine 10mg qd (8 a.m.), doxazosin 1mg qd (8 a.m.) and triazolam 0.25mg qd (10 p.m.).
Two days after discharge, the patient had two episodes of loss of consciousness, followed by
tonic contraction of the limbs lasting a few minutes, with residual, transient mental confusion. A CT
scan at the hospital emergency room excluded acute ischaemic or hemorrhagic events. The patient
refused an EEG. A third seizure occurred at night and was treated with i.v. diazepam. A further brain
CT excluded new acute events. As levofloxacin was suspected of playing a causal role in the seizures,
it was withdrawn. Therapy with phenytoin was instituted (100mg i.v. tid) but three days later had
to be stopped due to onset of muscular dyskinesia, and was replaced by levetiracetam (500mg
bd). One week after the last epileptic episode, cerebral magnetic resonance (MR) demonstrated
moderate cerebral atrophy, diffuse hypodensity of white matter, and bilateral alterations in the
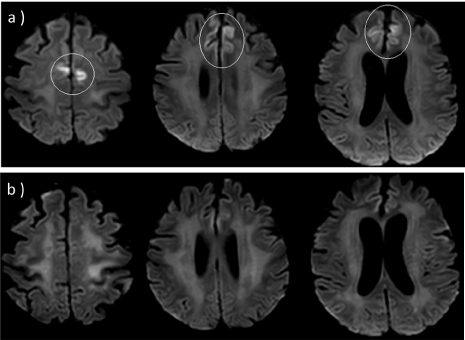
medial prefrontal cortex, compatible with cytotoxic oedema (Figure 1A). No significant cerebral
vessel stenosis were shown by angio-MR; nor did EEG show abnormal electrical activity. The
patient was discharged on levetiracetam (500mg bd) and re-evaluated 37 days later. On follow-up,
she remained seizure-free and MR showed complete regression of the morphological alterationspreviously appearing in the medial prefrontal cortex (Figure 1B).
To our knowledge, this finding has never been reported before and
needs to be briefly discussed. The changes seen in MR may have been
the consequence of the repeated epileptic attacks. Transient focal CT
and MR changes have been reported to occur after generalized tonicoclonic
seizures or status epilepticus, and attributed to local increases
in vascular perfusion and permeability and to massive release of
cytotoxic mediators, such as glutamate [1]. However, the direct causal
role of levofloxacin cannot be excluded. Fluoroquinolones are known
to produce excitatory CNS effects, which may result in adverse
psychiatric and neurologic reactions. Within this class of drugs,
levofloxacin has demonstrated a low epileptogenic potential [2]. In
addition, a literature review published in 2009 reported only six cases
of seizures associated with levofloxacin [3] and only three more have
been published since then [4-6]. Old age is a recognised risk factor for
fluoroquinolone-induced seizures. Our patient was very old (95), but
apparently she did not have any other predisposing conditions, such
as electrolyte imbalance, renal dysfunction, interacting drugs (e.g.,
NSAIDs, theophylline) or pre-existing seizure disorders.
Mechanisms proposed to explain fluoroquinolone neurotoxicity
include antagonism at the inhibitory GABAA receptor and synergism
at the excitatory NMDA receptor. The “GABA hypothesis” is
weakened by the evidence that fluoroquinolones have low affinity
for GABAA receptors. In particular, it has been estimated that
levofloxacin at concentrations expected in the human brain after
therapeutic doses occupies 1.1% of GABAA receptors, so that can
hardly exert clinically significant inhibition [7]. Instead, enhancement
of the excitatory glutamate effect produced by fluoroquinolones in
rat hippocampus slices [8] occurred at concentrations (0.5-1 μM)
compatible with levofloxacin-free concentrations expected in the
human brain (0.065 μM) [7]. In addition, quinolones reduced the
threshold for seizures by electroshock induced in a murine model and
their proconvulsive activity was completely blocked by an NMDA
receptor antagonist (MK-801) [9]. With this background, it seemsreasonable to suggest that fluoroquinolones can induce neurotoxicit mainly through a glutamatergic effect. If this is so, the MR cytotoxic
alterations seen in our patient after repeated seizures may have been
due to a combination of excessive neuronal discharge and the direct
glutamatergic effect of levofloxacin. Further studies are needed to
clarify the relative contribution of the two mechanisms.
Figure 1
Figure 1
Axial Diffusion Weighted Imaging (DWI): a) small symmetrical hyperintense areas in medial region of superior frontal circumvolution, corresponding to
medial prefrontal cortex area. Lesions show low values on Apparent Diffusion Coefficient (ADC) map, characteristic of cytotoxic oedema; b) follow-up MR images
37 days later, showing complete resolution of cytotoxic cortical lesions and regular homogeneous intensity of DWI signal along cortical layer in medial prefrontal
cortex on both sides.
Result
Acute cerebrovascular events were excluded. MR images obtained 7 days after the onset of seizures showed bilateral alterations in the medial prefrontal cortex, compatible with cytotoxic oedema. Thirtyseven days later, such alterations had disappeared.
Conclusions
Cytotoxic oedema may have been produced by massive glutamate release during epileptic episodes. However, a glutamatergic effect of fluoroquinolones cannot be excluded and deserves further study.
References
- Kim JA, Chung JI, Yoon PH, Kim DI, Chung TS, Kim EJ, et al. Transien MR signal changes in patients with generalized tonicoclonic seizures or status epilepticus: periictal diffusion-weighted imaging. Am J Neuroradiol 2001; 22: 1149-1160.
- Tome´ AM, Filipe A. Quinolones. Review of psychiatric and neurological adverse reactions. Drug Saf. 2011: 34: 465-488.
- Bellon A, Perez-Garcia G, Coverdale JH, Chacko RC. Seizures associated with levofloxacin: case presentation and literature review. Eur J Clin Pharmacol. 2009; 65: 959-962.
- Mazzei D, Accardo J, Ferrari A, Primavera A. Levofloxacin neurotoxicity and non-convulsive status epilepticus (NCSE): a case report. Clin Neurol Neurosurg. 2012; 114: 1371-1373.
- Gervasoni C, Cattaneo D, Falvella FS, Vitiello P, Cheli S, Milazzo L, et al. Levofloxacin-induced seizures in a patient without predisposing risk factors: the impact of pharmacogenetics. Eur J Clin Pharmacol. 2013; 69: 1611-1613.
- Famularo G, Pizzicannella M, Gasbarrone L. Levofloxacin and seizure: what risk for elderly adults? JAGS. 2014; 62: 2018-2019.
- Kim J, Ohtani H, Tsujimoto M, Sawada Y. Quantitative comparison of convulsive activity of combination of twelve fluoroquinolones with five nonsteroidal antiinflammatory agents. Drug Metab Pharmacokinet. 2009; 24: 167-174.
- Schmuck G, Schürmann A, Schlüter G. Determination of the excitatory potencies of fluoroquinolones in the central nervous system by an in vitro model. Antimicrob Agents Chenother. 1998; 42: 1831-1836.
- Williams PD, Helton DR. The proconvulsive activity of quinolone antibiotics in an animal model. Toxicol Lett. 1991; 58: 23-28.