Case Report
An Unusual Case of a Neonatal Heart Murmur (Cardiac Tumour Presenting as a Heart Murmur in a Neonate)
Oluwasomidotun Idowu1*, Tarak Desai2 and Anjum Gandhi1
1Good Hope Hospital, Birmingham, UK
2/Birmingham Children’s Hospital, Birmingham, UK
*Corresponding author: Oluwasomidotun Idowu, Good Hope Hospital, Birmingham, UK
Published: 11 Aug, 2016
Cite this article as: Idowu O, Desai T, Gandhi A. An
Unusual Case of a Neonatal Heart
Murmur (Cardiac Tumour Presenting as
a Heart Murmur in a Neonate). Ann Clin
Case Rep. 2016; 1: 1060.
Abstract
A term neonate was found to have a harsh ejection systolic murmur on routine examination. The
murmur was heard loudest at the left sternal edge, radiating throughout the precordium. Despite
being clinically well with normal pulses and saturations, an early echocardiogram was arranged
as the murmur sounded pathological. Echocardiogram revealed moderate stenosis of the right
ventricular outflow tract (RVOT). Repeat ECHO two weeks later showed dramatic increase in
narrowing of the RVOT prompting urgent tertiary referral. Radiological investigations revealed a
tumour within the anterior mediastinum compressing the RVOT.
Excision of the tumour showed that it arose from within the myocardium. A histological diagnosis
of cardiac fibroma was made.
Primary paediatric cardiac tumours are extremely rare. Much of the information we know is derived
from case reports. Here, we aim to add to that reservoir, and draw attention to a very uncommon
cause of very a common sign (heart murmurs).
Background
Primary paediatric cardiac tumours are extremely rare with a frequency of 0.0017 to 0.02% [1].
Due to their rarity, much of the clinical information we have is derived from case reports. The lack
of knowledge in this area combined with the variability in presentation of cardiac tumours makes
them a challenge to diagnose.
With this case report we aim to add to the clinical knowledge of cardiac tumours. In addition,
we want to draw attention to this very rare cause of heart murmurs.
Case Presentation
A term neonate, fit and well, was found to have a heart murmur during a routine
neonatal check on day 1 of life. Father was known to have a heart murmur as a child
that had resolved spontaneously. There was no significant family history of heart
disease. Maternal serology during pregnancy and antenatal scans were normal.
On examination, the child was pink, warm and well perfused with saturations of 98% on air. Heart
sounds I+II were normal with a harsh ejection systolic murmur (grade 3/6), loudest at the left sternal
edge but heard throughout the precordium. Femoral pulses were palpable bilaterally with normal
character. Respiratory examination was unremarkable with no signs of increased work of breathing.
No respiratory distress, sweating or colour change was observed during bottle-feeding. Abdomen
was soft, non- tender, no organomegaly, and bowel sounds present. As per unit protocol, the baby
was observed overnight on the postnatal ward and reassessed the following morning. Failure of the
murmur to resolve over a 24 hour period led to further assessments, in line with hospital guidelines,
to identify the aetiology. Pre and post-ductal saturations (98% and 96% respectively) and pre
and post-ductal blood pressure (78/52 mm and 97/46 mm respectively) were checked and were
normal. An electrocardiogram (ECG) was also done and reported as normal.
The guideline recommends arranging a review in the paediatric clinic in 2 to 3 weeks if the
murmur persists but the baby is clinically normal and investigations unremarkable. Although
this would have been the recommended action, a clinical decision was made to undertake an
echocardiogram in view of the character and grade of murmur, which suggested the possibility of a
pathologic murmur secondary to a congenital heart defect.
The echocardiogram revealed normal situs, normal atrioventricular and ventriculo- arterial connections. Atrial septum and ventricular septum were intact and arterial duct was not patent. No left ventricular outflow obstruction was noted. However, turbulence and acceleration of flow at the right ventricular outflow tract (RVOT) was detected, suggesting narrowing of the RVOT. The RVOT Doppler was 3.4m/sec leading to a diagnosis of moderate pulmonary stenosis.
Although the echocardiographic diagnosis by itself did not prompt an early review in view of the age of the child, an earlier follow up of two weeks was arranged.
At follow up, the patient remained clinically well and asymptomatic. Saturations were 97% on air and cardiac and systemic examination was as before. However, repeat echocardiogram revealed that the narrowing of the right ventricular outflow tract had increased significantly with a RVOT Doppler of more than 4m/sec. Moreover, the right ventricular out flow tract was noted to be narrow and elongated with an abnormal squashed appearance (Figure 1-3). The accelerated change in the degree of stenosis combined with the unusual appearance of the RVOT, led to the patient being referred to a tertiary paediatric cardiology centre for urgent assessment.
Figure 1
Figure 1
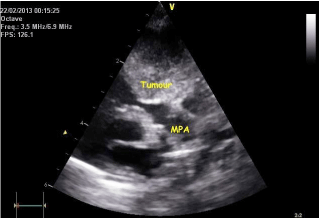
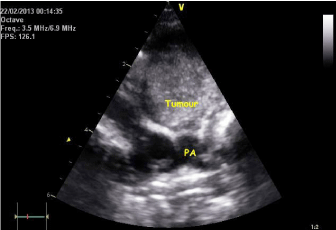
Echocardiogram still image (modified parasternal view) showing tumour encroaching the main pulmonary artery.
Figure 2
Figure 2
Echocardiogram still image (modified parasternal view) showing the tumour situated outside the right ventricle and compressing the right ventricular outflow tract (RVOT).
Figure 3
Figure 3
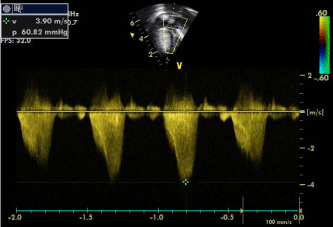
Doppler echocardiography showing acceleration of flow in the RVOT with a velocity of 3.9 m/s because of narrowing.
Treatment
In light of the accelerated growth of the mass and failure to determine its origin and primary source, surgical exploration and excision of the tumour was decided with patch repair of the right ventricular outflow tract.
The procedure involved creating a median sternotomy in order to gain access and visualization. A subtotal thymectomy was then performed. Cardiopulmonary bypass was established between the ascending aorta and bicaval cannulation. Upon establishment, the aorta was cross-clamped and the heart arrested with cold crystalloid cardioplegia administered via the aortic root. The RVOT was identified and the tumour completely excised using sharp dissection and diathermy. Consequently, a large defect remained in the RVOT, immediately beneath the pulmonary valve. This was repaired using a patch of bovine pericardium. Finally, the heart was de-aired and the cross clamp removed with a vent on the aortic root. The heart was weaned from cardiopulmonary bypass, following rewarming, in sinus rhythm. No inotropic support was required.
Findings from the procedure revealed that the thymus was normal in appearance and the mass clearly arose from within the myocardium of the RVOT with the left arterial descending artery to the left and a marginal branch of the right coronary artery to the right of the mass. The tumour was noted to have a bosselated surface, to be well demarcated and closely adherent to the myocardium at the edges with no true plane between them. Histology report revealed that the mass was a cardiac fibroma.
Outcome and follow-up
Surgery was successful and on post-op review the child was fit and well. On examination he was pink and well perfused. Saturations of 98% and heart sounds I+II present with no added sounds. Echocardiogram showed good biventricular function with laminar flow in the right ventricular outflow tract. No compression was detected and there was no evidence suggestive of cardiac tumour or pericardial effusion.
Discussion
Primary cardiac tumours within the paediatric community are rare, with a prevalence of 0.0017 to 0.02% [1]. They are particularly uncommon in infancy with a handful of case reports describing them. Most of the tumours are benign with rhabdomyomas, fibromas and teratomas accounting for the majority of cases [1].
Due to the rarity and variability in presentation of cardiac tumours, they pose a clinical challenge to diagnose. Heart murmurs on the other hand are very common in children and infants (including neonates) and are often innocent or secondary to congenital heart disease. It is not usual for a diagnosis of cardiac tumour to be considered by the clinician who on auscultation discovers that a child has a heart murmur.
Presentation depends on the size and location of the mass rather than the histologic type as they have a tendency to produce obstruction of the inflow or outflow tracts or induce arrhythmias [2-5]. Cyanosis, breathlessness and ventricular arrhythmias have all been described as presenting features [6]. The symptoms and signs created by the cardiac tumour can also be used to clinically speculate whether it is arising from the right or left hand side of the heart. Right-sided cardiac tumours are likely to present with associated syncope, cyanosis, vena-cava shunt syndrome, pulmonary embolisms and/or pericardial effusions [7]. On the other hand, left sided tumours are likely to result in embolisation causing transient ischaemic attacks, seizures, and/or peripheral vascular and/or cerebral vascular accidents [2].
The diagnosis of cardiac tumours is easier in patients presenting with the above mentioned symptoms as it often leads to a thorough assessment including cardiac imaging. It is a much greater challenge in the asymptomatic child where the clinical condition is unlikely to trigger prompt cardiac imaging. Worryingly, sudden death is recognized as a presenting feature suggesting that children with cardiac tumours may have no clinical clues before presenting with catastrophic collapse. Certainly fibromas have been recognized to present with significant cardiac compromise including sudden death. A very similar, but symptomatic case (with cyanosis and progressive dyspnoea), has been described involving an 8-month old infant with RVOT obstruction secondary to a large cardiac fibroma (5 x 4.5cm) [6].
In our case, although the baby was asymptomatic, the pathologic nature of the murmur and the age at presentation prompted an early echocardiographic assessment and early diagnosis.
Echocardiogram, CT and MRI scans are first line in providing diagnostic information for evaluation of cardiac tumours. Echocardiogram allows for a dynamic, structural view of the heart aiding in identifying any masses alongside evaluating the mobility of the tumour. In addition, it allows one to view the effect that the tumour is having upon the surrounding structures [3]. MRI is generally a better imaging modality than CT although both are of value. Diagnosing the type of tumour can be more reliably made with a MRI scan as the T1 and T2 weighted sequences can give clues about the microenvironment within the tumour [3,5,8-10]. PET scan, coronary angiogram and transvenous biopsy are also useful diagnostic tests depending on the clinical context [5]. Histological diagnosis however, remains the gold standard for determining the type of tumour.
Fibromas are the second most common cardiac tumours in infancy but rank first in surgical series [11]. They often arise within the ventricular muscle, most commonly the ventricular septum although can also arise from within the free walls of the ventricles [7]. Unlike rhabdomyomas, they do not spontaneously resolve [12]. They can cause obstruction to blood flow, valvular dysfunction, arrhythmias, syncope and/or sudden death in 10-30% of cases [13-15]. Of arrhythmias associated with cardiac fibromas, ventricular tachycardia accounts for 50% [13].
Although for many cardiac tumours, especially rhabdomyomas, watchful waiting is an acceptable management option, surgery, if needed, is usually safe and definitive [16]. However, watchful waiting is not applicable to fibromas which are not known to undergo spontaneous regression [12]. In fact, in the case of cardiac fibromas, surgery should be offered early because of their tendency to grow fast and cause severe haemodynamic complications. Ideally complete surgical resection with chamber reconstruction using pericardial patch should be done and this was achieved in our case. However, excellent midterm results have been achieved even with partial resection and hybrid procedures using BT shunt or bidirectional cavopulmonary connection to offload the right ventricle [13,17,18]. In cases with persistent arrhythmias in the setting of a non- resectable tumour, ICD implantation may be needed. Rarely the only treatment option possible may be cardiac transplantation [3,19].
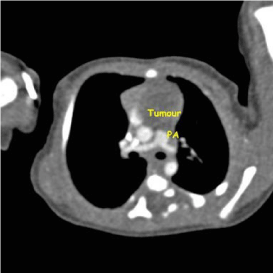
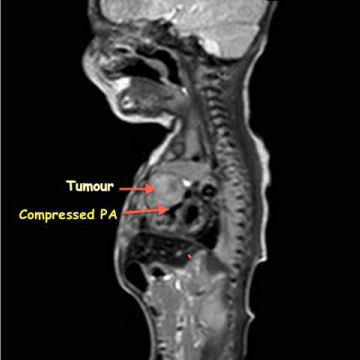
Figure 4
Figure 5
Learning Points
1. Although rare, cardiac tumours should be considered in the differential diagnosis of paediatric cases presenting with unexplained murmurs, heart failure or arrhythmia.
2. Once a diagnosis of a cardiac tumour is suspected prompt and thorough assessment using a variety of cardiac imaging modalities including echocardiography, CT and MRI scanning should be arranged in a tertiary cardiac centre. This is important as some of the cardiac tumours can grow rapidly with significant development of cardiac compromise and even sudden death.
3. Cardiac tumours have the potential to cause serious cardiac compromise and death but if diagnosed in a timely manner and managed appropriately with surgical intervention the prognosis is good in most cases.
Acknowledgements
The author would like to thank Valerie Carter for her administrative support towards the development of this manuscript. In addition, the author would also like to thank the family for giving their consent for this manuscript to be published in order to raise awareness about this rare condition.
Footnotes
Contributors: All authors contributed to the case report and agreed on the final manuscript. OI was involved in both drafting and revision of the manuscript. TD revised the manuscript and provided the accompanying images. AGcontributed to drafting and revision of the manuscript as well supervising its development.
References
- Uzun O, Wilson DG, Vujanic GM, Parsons JM, De Giovanni JV. Cardiac tumours in children. Orphanet. Journal of Rare Diseases. 2007; 2: 11.
- Cordinha C, Pereira A, Silva PV, Dionisio T, Martins P, Sousa G, et al. Asymptomatic cardiac tumor in a child: an incidental diagnosis. Revista Portuguesa De Cardiologia. 2011; 30: 795-797.
- Gaash WH, Vander Salm TJ. Cardiac tumors. In: Post TW, editor. UpToDate. Waltham, MA: UpToDate; 2015.
- Sheu C-C, Lin S-F, Chiu C-C, Lee C-S, Lee S, Chai C-Y, et al. Left atrial sarcoma mimicking obstructive pulmonary disease. Journal of Clinical Oncology. 2007; 25: 1277-1279.
- Vander Salm TJ. Unusual primary tumors of the heart. Seminars in thoracic and cardiovascular surgery. 2000;12: 89-100.
- Wu JR, Chiu CC, Lin YT, Dai ZK, Lin HJ. Cyanosis caused by a huge obstructive right ventricular fibroma. Japanese Heart Journal. 2000; 41: 239-243.
- Burke A, Virmani R. Pediatric heart tumors. Cardiovascular Pathology. 2008; 17: 193-198.
- Kaminaga T, Takeshita T, Kimura I. Role of magnetic resonance imaging for evaluation of tumors in the cardiac region. European Radiology. 2003; 13: L1-L10.
- Gulati G, Sharma S, Kothari SS, Juneja R, Saxena A, Talwar KK. Comparison of echo and MR1 in the imaging evaluation of intracardiac masses. Cardiovascular and Interventional Radiology. 2004; 27: 459-469.
- Araoz PA, Eklund HE, Welch TJ, Breen JF. CT and MR imaging of primary cardiac malignancies. Radiographics. 1999; 19: 1421-1434.
- Basso C, Valente M, Poletti A, Casarotto D, Thiene G. Surgical pathology of primary cardiac and pericardial tumors. European Journal of Cardio-Thoracic Surgery. 1997; 12: 730-738.
- Kohut J, Ladzinska JK, Szydlowski L, Smolenska-Petelenz J, Giec-Fuglewicz G, Pajak J. The diagnosis, clinical course and follow-up of children with cardiac tumours - a single- centre experience. Kardiologia Polska. 2010; 68: 304-309.
- Miyake CY, Del Nido PJ, Alexander ME, Cecchin F, Berul CI, Triedman JK, et al. Cardiac Tumors and Associated Arrhythmias in Pediatric Patients, With Observations on Surgical Therapy for Ventricular Tachycardia. Journal of the American College of Cardiology. 2011; 58: 1903-1909.
- Stratemann S, Dzurik Y, Fish F, Parra D. Left ventricular cardiac fibroma in a child presenting with ventricular tachycardia. Pediatric Cardiology. 2008; 29: 223-226.
- Ottaviani G, Rossi L, Ramos SG, Matturri L. Pathology of the heart and conduction system in a case of sudden death due to a cardiac fibroma in a 6-month-old child. Cardiovascular Pathology. 1999; 8: 109-112.
- Padalino MA, Basso C, Milanesi O, Vida VL, Moreolo GS, Thiene G, et al. Surgically treated primary cardiac tumors in early infancy and childhood. Journal of Thoracic and Cardiovascular Surgery. 2005; 129: 1358-1363.
- Lee C, Kim S-J, Kim YM. Cardiac Fibroma in an Infant: Complete Resection After aBlalock-Taussig Shunt as Initial Palliation. Annals of Thoracic Surgery. 2010; 90: 1011-1014.
- Freire G, Jureidini S, Schowengerdt KO, Rahimi B, Tinker K, Fiore AC. Hybrid Procedure for Obstructive Neonatal Left Ventricular Tumors. Annals of Thoracic Surgery. 2010; 90: 2057-2059.
- Becker AE. Primary heart tumors in the pediatric age group: A review of salient pathologic features relevant for clinicians. Pediatric Cardiology. 2000; 21: 317-323.